Background: Pulmonary arterial hypertension (PAH) and chronic thromboembolic pulmonary hypertension (CTEPH) are characterised by right ventricular (RV) pressure overload, dilatation, and RV-dysfunction. These are key determinants of the prognosis for patients with PAH. Although riociguat improves exercise capacity and haemodynamics, there is limited prospective data on its impact on the right heart. This prospective study aimed to evaluate the effect of riociguat on right heart size, function, haemodynamics, exercise capacity, and safety in patients with PAH or CTEPH.
Methods: RIVER II was a prospective, open-label, single-centre, phase IV trial. The primary endpoint was the change in right atrial (RA) size, as measured by echocardiography from baseline to week 24. The secondary endpoints included changes in additional echocardiographic indices, invasive haemodynamics, six-minute walk distance (6MWD), functional class (WHO-FC), N-terminal pro-brain natriuretic peptide (NT-proBNP), quality of life, and safety.
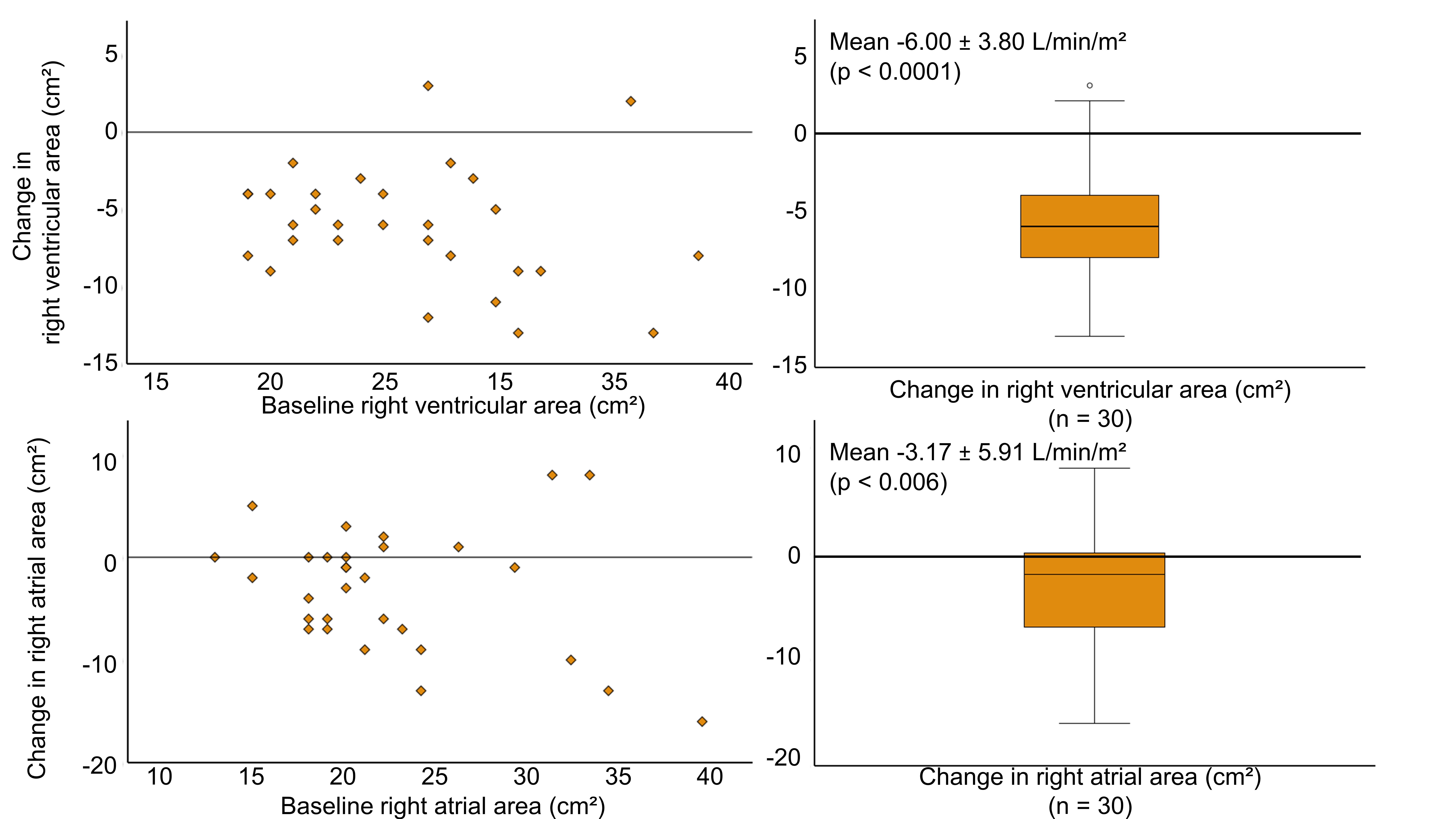
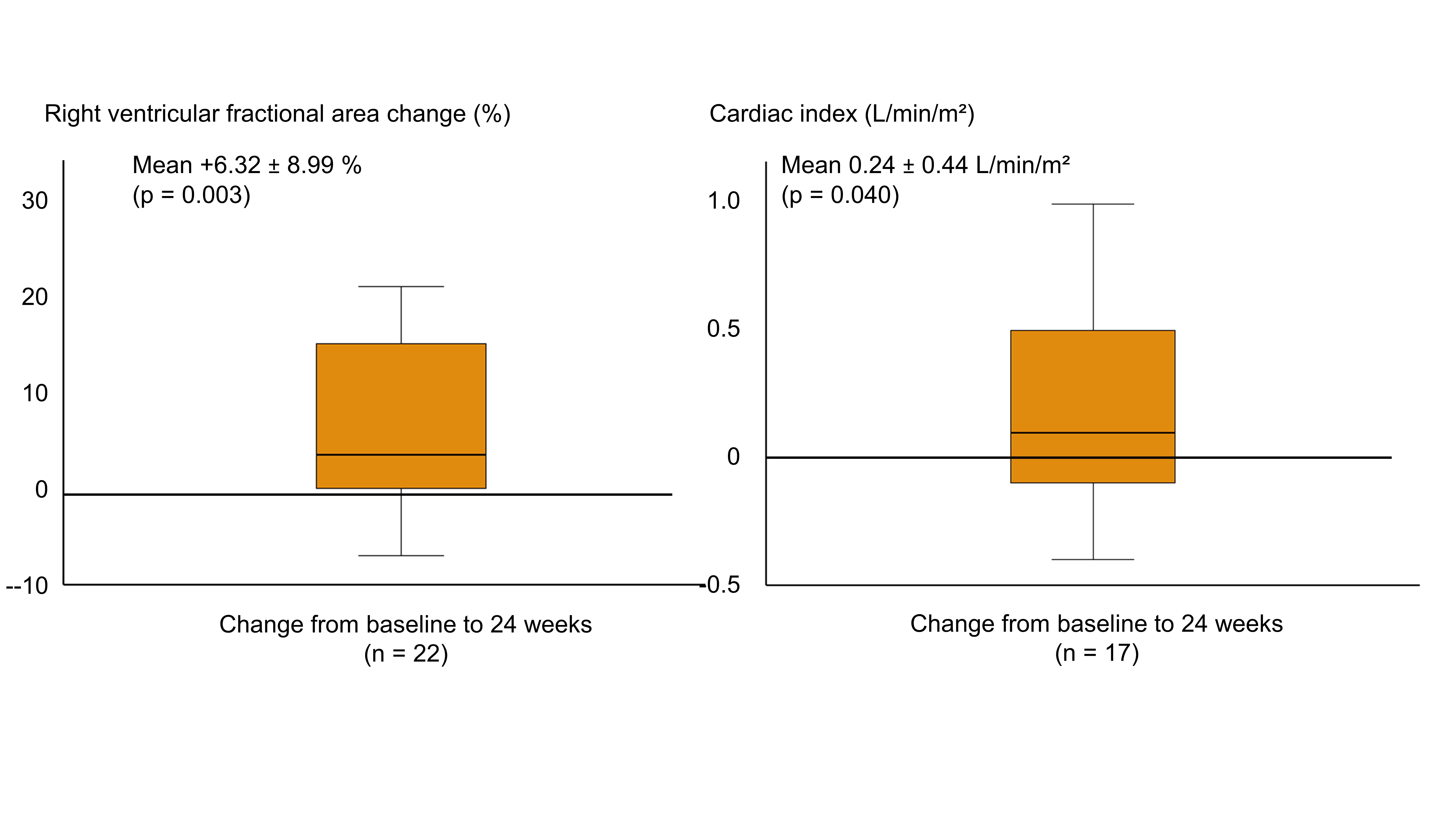
Results: Thirty patients (mean age 61.2 ± 14.5 years; 76.7% male) were enrolled, of whom 24 switched from phosphodiesterase type 5 inhibitors (PDE5i) to riociguat. At baseline, participants demonstrated significant right-heart dilation (RA: 22.50 ± 6.14 cm²; RV: 26.10 ± 5.50 cm²) and reduced RV function. Treatment with riociguat (MK-4836) led to a significant improvements in the primary endpoints with a reduction in mean RA area (Δ −3.17 ± 5.91 cm², p = 0.006) and mean RV area (Δ −6.00 ± 3.80 cm², p < 0.001). Secondary endpoints, including RV fractional area change (Δ +6.32% ± 8.99%; p = 0.003), 6MWD (Δ +41.00 m ± 57.94; p = 0.01) and WHO-FC (improvement in 56.7% of patients; p < 0.001), were also significantly improved. In the 17 patients for whom haemodynamic measurements were available, cardiac index increased (Δ +0.24 ± 0.44 L/min/m²; p = 0.040) and cardiac output improved (Δ +0.41 ± 0.81 L/min; p = 0.054), while pulmonary vascular resistance decreased (Δ −1.04 ± 2.89 WU; p = 0.158). NT-proBNP and quality-of-life scores showed no significant changes. Safety was consistent with the known profile of riociguat. There were no deaths during the study.
Conclusion: This is the first prospective trial to show that riociguat led to significant reverse remodelling of right heart size and function within 24 weeks of treatment. Improvement of the right ventricle was associated with significant improvement of secondary endpoints, including exercise capacity and functional class. The therapy was well tolerated, with no new safety concerns emerging. Despite the study's limitations (single-centre, open-label design, small sample size, and early termination), the findings support the use of riociguat as an effective treatment for improving right ventricular (RV) geometry and performance. Larger, controlled trials are warranted.