Background
Accurate identification of acute coronary occlusion (ACO) in chest pain patients remains challenging, as standard ST-segment elevation myocardial infarction (STEMI) criteria frequently fail to detect ACO—present in up to one in four non-STEMI cases. Artificial intelligence (AI)-enhanced ECG analysis may improve diagnostic accuracy, particularly in tachycardic patients, where ST-segment deviations can result from artefacts, supply–demand imbalance, or true ACO. Differentiating these causes is crucial to guide emergency angiography versus arrhythmia management.
Objective
To assess the diagnostic performance of an AI-enhanced ECG model for detecting ACO in chest pain patients with tachycardia on admission ECG, compared with conventional STEMI criteria, in a metropolitan chest pain unit (CPU) cohort.
Methods
The AI model, built on a deep convolutional neural network, was previously trained and validated in international cohorts. For the present analysis, all consecutive tachycardic patients (>100 bpm) presenting with chest pain to the CPU between November 2018 and June 2021 were included. Clinical management followed current European Society of Cardiology recommendations, including the 0/1-hour high-sensitivity cardiac troponin T algorithm where applicable. The endpoint was the presence of ACO, adjudicated by an independent physician blinded to the AI model’s predictions, based on coronary angiography, biomarker kinetics, and echocardiography. Diagnostic performance was evaluated using receiver operating characteristic (ROC) analysis and compared with STEMI criteria. A predefined subgroup analysis distinguished between narrow (QRS <120ms) and broad (QRS ≥120ms) complex tachycardias.
Results
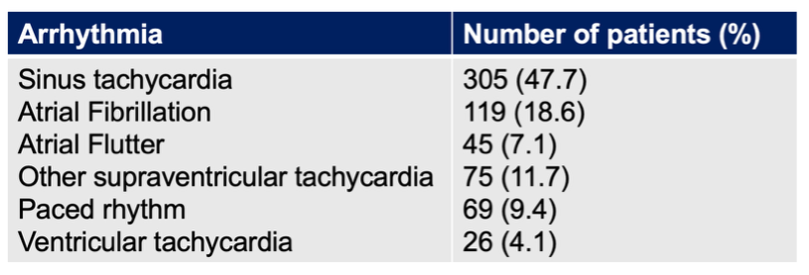
Among 4,440 CPU admissions, 639 patients presented with tachycardia and were included (mean age 56.3 ± 19.3 years; 60.6% male). Underlying tachycardia or tachyarrhythmia are summarized in figure 1. Forty-nine patients (7.7%) had broad complex tachycardia, while 590 (92.3%) had narrow complex tachycardia.
Figure 1
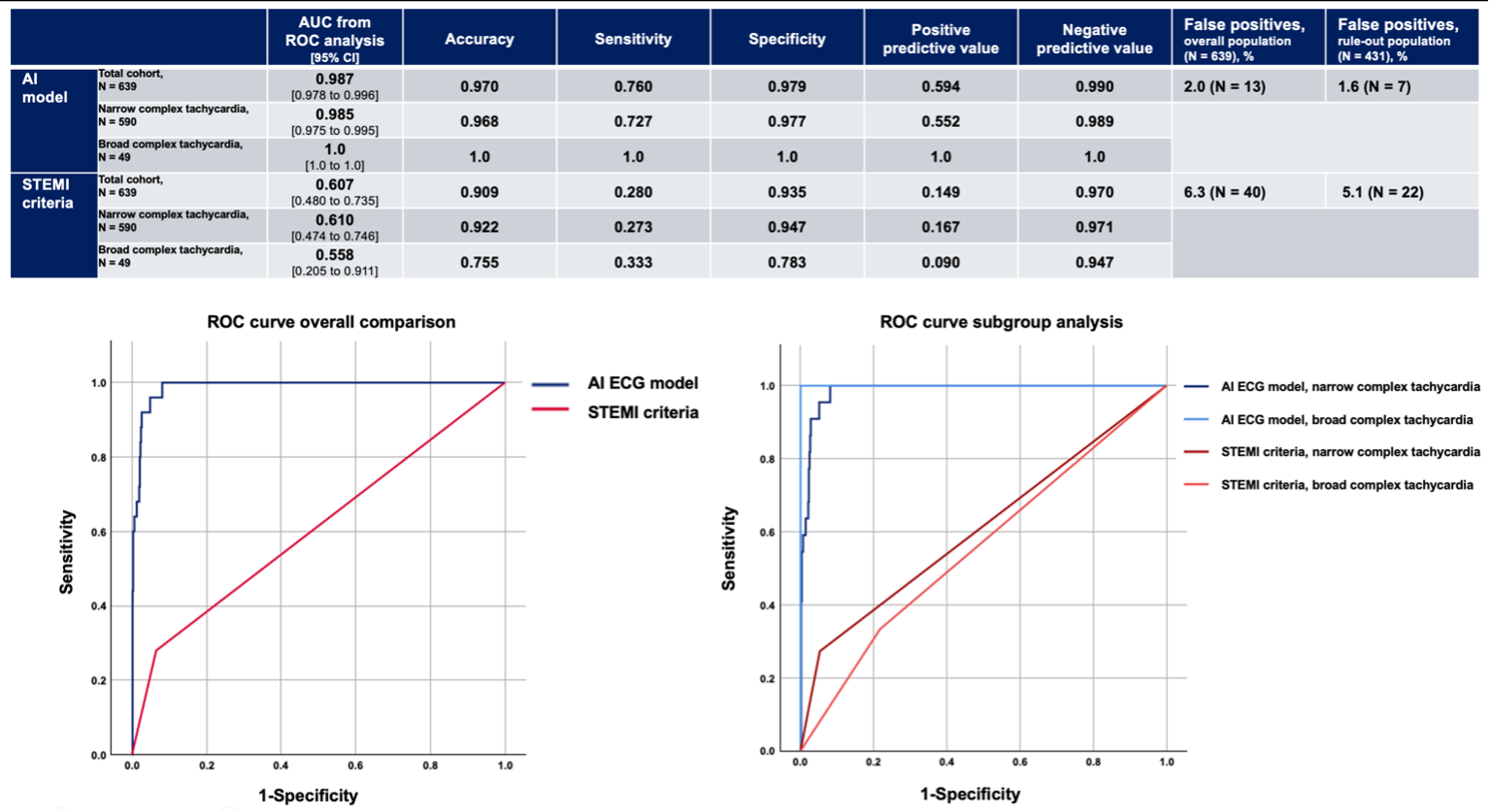
The overall prevalence of ACO was 3.9% (n = 25). STEMI criteria identified only 7 of these 25 cases (28%). In contrast, the AI-ECG model correctly detected 19 of 25 (76%) ACO. Across all diagnostic metrics—including AUC, accuracy, sensitivity, and predictive values—the AI model outperformed STEMI criteria, consistently across both tachycardia subgroups (Figure 2). In the low-risk rule-out population, the AI-ECG model yielded seven false positives, potentially avoiding fifteen unnecessary emergency angiographies compared with strict application of STEMI criteria.
Conclusion
In patients with chest pain and sinus tachycardia or tachyarrhythmia, conventional STEMI criteria insufficiently guide emergency coronary angiography, causing both missed occlusions and potentially avoidable procedures.
AI-enhanced ECG interpretation improved ACO detection and may enable more selective, clinically guided triage. Its strong negative predictive value highlights its potential for guiding decisions between primary arrhythmia management and emergency coronary angiography, warranting prospective validation.
Multipanel Figure 2