Background Pulsed field ablation (PFA) has recently emerged as a promising energy source for pulmonary vein isolation (PVI) in atrial fibrillation (AF) ablation. The integration of three-dimensional (3D) electroanatomical mapping allows detailed assessment of lesion completeness and atrial substrate characterization. Whether an additional high-density mapping catheter provides superior mapping accuracy compared to mapping performed solely with the ablation catheter remains uncertain.
Aims This study aimed to compare procedural efficiency and mapping effectiveness between PFA-guided AF ablation performed with a dedicated 3D mapping catheter and PFA using the ablation catheter itself for 3D map creation.
Methods A total of 200 consecutive patients undergoing first-time PFA for AF with a commercially available pentaspline PFA system were prospectively enrolled.
In 100 patients (two-catheter-group), high-resolution 3D maps of the left atrium were created using a separate mapping catheter both before and after PFA. PVI was verified in the post-ablation map, and additional lesions were delivered in case of PV reconnection or residual conduction. In the other 100 patients (single-catheter-group), 3D maps were created exclusively with the PFA ablation catheter following the same procedural protocol. In both groups, mapping was performed to identify low-voltage areas, followed by additional ablation when appropriate. Procedural parameters, mapping findings, PV reconnection rates, and complications were compared between groups.
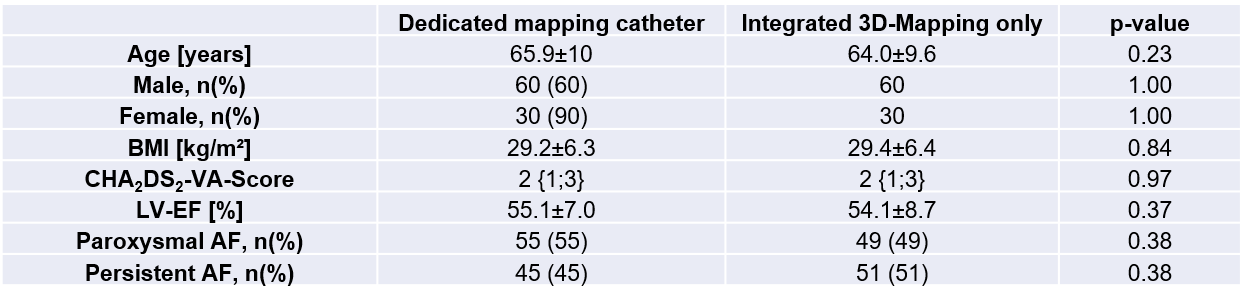
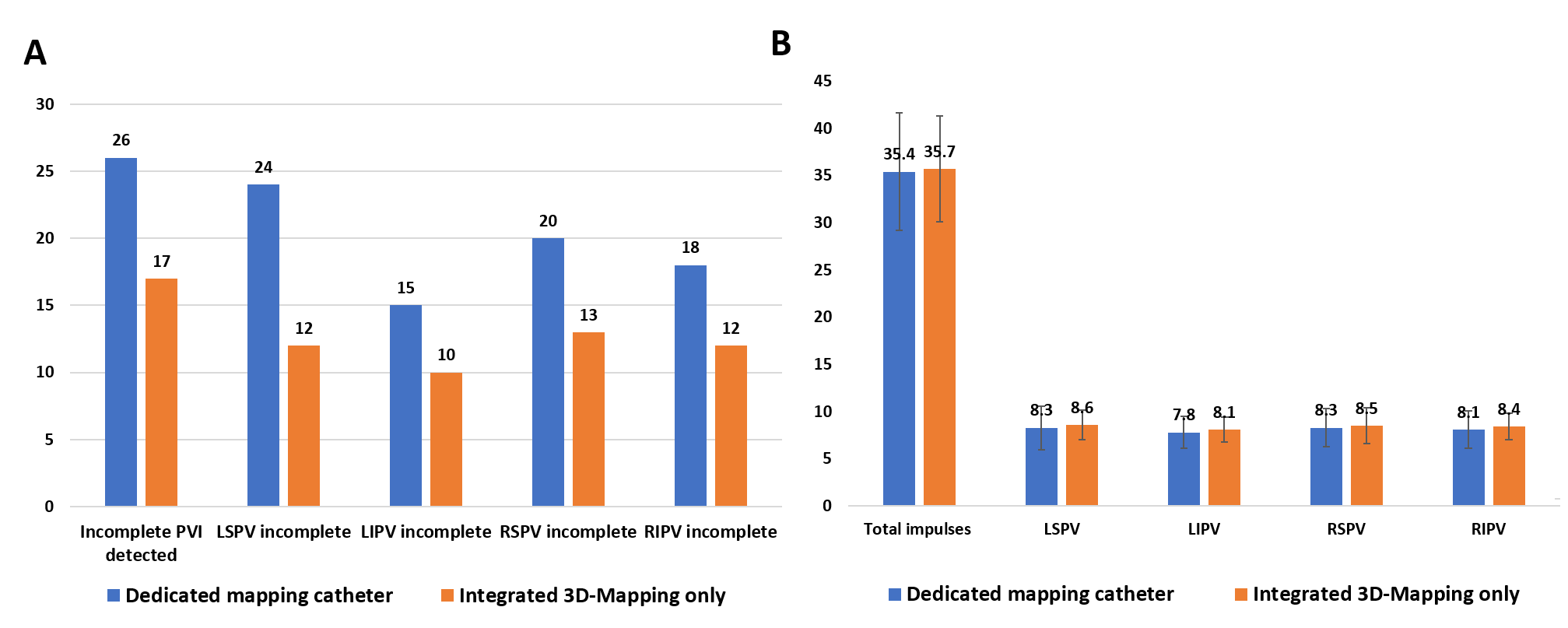
Results Baseline characteristics were comparable between groups (table 1). Mean procedural duration was modestly shorter in the integrated mapping group (46.9±16.9 minutes vs. 50.8±13.4 minutes; p=0.07), while fluoroscopy time was significantly longer when mapping was performed with the ablation catheter alone (11.5 ± 2.5 minutes vs. 7.9±3.3 minutes; p< 0.001). Total ablation impulses and fluoroscopy dose were comparable between groups (p=0.13 and p=0.78, respectively). Incomplete PVI was observed in 26% of patients in the dedicated mapping–catheter group and in 17% of patients in the integrated mapping group (p=0.12). The distribution of incomplete PVI sites and the need for additional ablation beyond PVI did not differ significantly between groups (figure 1). Presence of low voltage was similar (14% vs. 10%; p = 0.36), and procedural complications were infrequent (1 femoral pseudoaneurysm in the dedicated mapping catheter group, no complications in the device mapping group) and comparable in both groups.
Conclusions PFA-guided AF ablation using only the ablation catheter for 3D mapping provides procedural and electrophysiological results comparable to those obtained with an additional dedicated mapping catheter. While omitting the mapping catheter slightly reduces procedure duration and resource use, mapping quality, PVI verification, procedural safety and low voltage detection remain equally effective. Thus, integrated 3D mapping with the ablation catheter represents an efficient and reliable strategy for PFA-guided atrial fibrillation ablation.