Background: Transcatheter aortic valve intervention (TAVI) has been established as the preferred method for most elderly patients suffering from severe symptomatic aortic valve stenosis. For many years only one balloon-expandable aortic valve (BEV) was available. Recently, a second one, the Meril Myval transcatheter heart valve (THV) series, became available. We aimed to evaluate efficacy and safety of this new BEV in clinical practice.
Methods and results: We performed a matched-pairs comparison between the BEV Meril Myval Octacor®, the BEV Edwards Sapien-3®/Ultra® and the self-expanding (SEV) Medtronic Evolut® THVs.
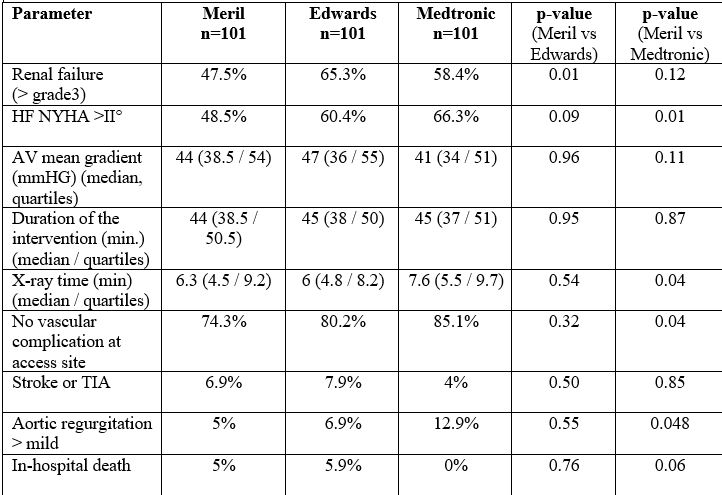
Results: Out of more than 3500 TAVI patients treated at our institution, 101 patients intended to be treated with the Meril THV were compared to equal numbers of matched patients treated with Medtronic and Edwards THVs. Only patients treated by transfemoral access were included. Matching parameters were age, sex, logistic EuroScore2 and valve size. Patients were observed until hospital discharge. Median age of patients was 83 years. Matching resulted in a similar distribution of base-line characteristics.
Baseline and interventional characteristics as well as in-hospital results are shown in the table.
All combined endpoints (feasibility, early safety and clinical events according to the VARC-3 definitions) were not statistically different between the three THVs.
Conclusions:
Our single-centre, real-world, matched-pairs comparison between the Meril Octacor®, the Medtronic Evolut® and the Edwards Sapien3®/Ultra® THVs showed comparable interventional success and safety rates, as well as similar clinical results until hospital discharge.