Background
Transthyretin amyloid cardiomyopathy (ATTR-CM) is an infiltrative disease that stiffens the myocardium, leading to heart failure, conduction disturbances, and arrhythmias. Historically considered untreatable, guideline recommendations for primary prevention of sudden cardiac death with implantable cardioverter-defibrillators have been conservative. With advances in diagnostic modalities and disease-specific therapies prolonging survival, the prognostic relevance of ventricular arrhythmias merits re-evaluation. We aimed to determine the frequency of ventricular arrhythmias in ATTR-CM and evaluate their association with all-cause mortality.
Methods
We prospectively enrolled 282 patients with confirmed ATTR-CM from two tertiary amyloidosis centers between 2017 and 2025. All participants underwent Holter ECG monitoring alongside detailed clinical, laboratory, and imaging assessments. Vital status was ascertained through structured telephone follow-up and review of the civil death registry. Ventricular arrhythmias were categorized as premature ventricular contractions (PVCs), accelerated idioventricular rhythm (IVR), non-sustained ventricular tachycardia (nsVT) and sustained ventricular tachycardia (sVT). Survival differences were evaluated using Kaplan-Meier analysis and log-rank tests. Predictors of all-cause mortality were sought using multivariable Cox regression models.
Results
This unselected cohort encompassed 282 ATTR-CM patients. Ventricular arrhythmias were frequent, occurring in 108 of 260 patients with complete Holter data (41%). Specifically, 28% (n=78) showed nsVTs and 23% (n=66) IVR, whereas sVTs were rare (0.7%, n=2). PVCs were present in 92% (n=261) of patients, with a median number of 410 (107-1804) PVCs per patient, corresponding to a median VC burden of 0.5%. Relevant pauses (>2s) were detected in 30% (n=78).
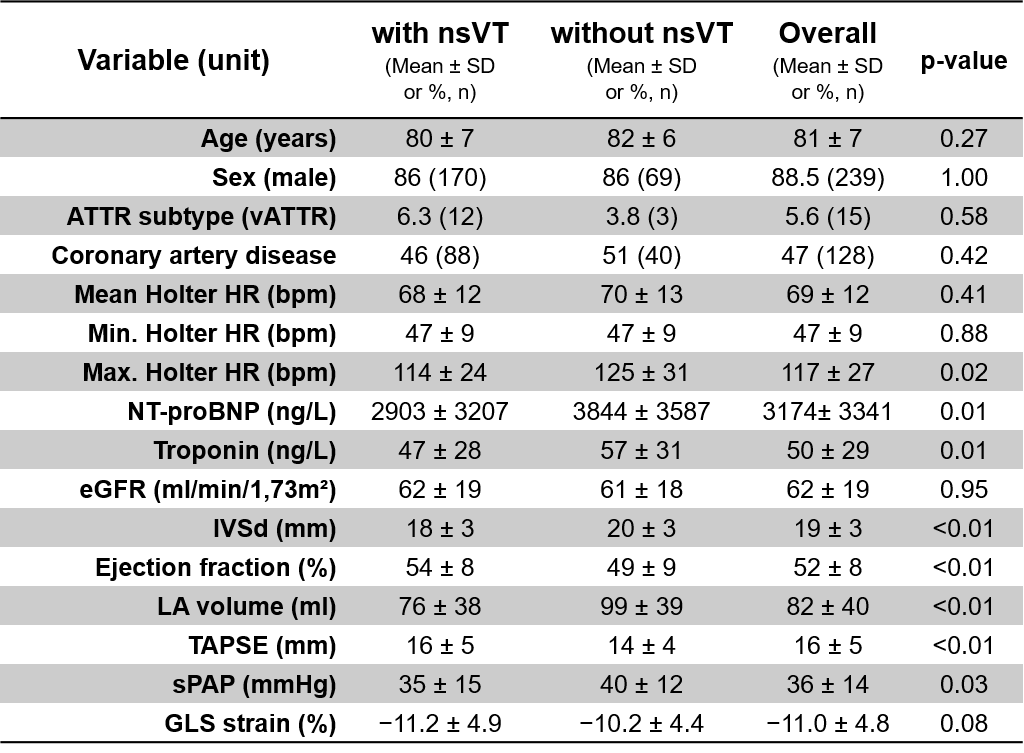
Patients with nsVTs had more advanced disease stage, reflected by higher NT-proBNP and troponin levels and worse imaging parameters, including lower ejection fraction (LVEF) and tricuspid annular plane systolic excursion (TAPSE), greater septum diameter (IVSd), and enlarged left atria (Table 1). Despite this, nsVT was not associated with an increased risk of all-cause mortality (23% vs. 16%, p=0.387, mean follow-up time 998 [85%-CI 902-1094] days). By contrast, one of the two patients with sVTs died during follow-up (50% vs. 18%, p=0.016).
In a multivariable Cox-regression model (including nsVTs, pauses >2 seconds, age, NTproBNP, Troponin, GFR, LVEF, IVSd, TAPSE, IVR); age, NTproBNP, GFR and TAPSE were retained in the final model as predictors for mortality (χ²=34, DF=4, p<0.001).
Conclusion
In this cohort of ATTR-CM patients, ventricular arrhythmias were a common finding during Holter monitoring and reflected advanced myocardial involvement. Frequent nsVTs, although associated with markers of disease severity, did not translate into higher all-cause mortality. Sustained VTs occurred rarely but portended a worse prognosis, suggesting that only a small subset of ATTR-CM patients are at risk of sudden arrhythmic death. Larger studies with long-term follow-up in ATTR-CM patients are warranted to clarify which individuals might benefit from prophylactic defibrillator therapy.