Background: Atrial fibrillation (AF) and chronic kidney disease (CKD) are closely intertwined, sharing bidirectional causal pathways and common risk factors. Whether this interplay is mediated by left atrial hypertension and whether renal function modifies the relationship between left atrial pressure and AF recurrence after pulmonary vein isolation (PVI) remain unclear.
Methods: We performed a two-center retrospective study of consecutive first-time PVI procedures under moderate sedation. Mean left atrial pressure (LAPm) was measured immediately after transseptal puncture (TSP). Clinical, echocardiographic, and laboratory data were collected without age, sex, or comorbidity restrictions. Associations between LAPm and covariates were examined using Spearman correlations. To test whether kidney function modifies the relationship of LAPm and AF recurrence, we performed a general linear model (ANCOVA) with recurrence status, eGFR, and their interaction, and a multivariable logistic regression for AF recurrence, including LAPm, eGFR, and key clinical covariates.
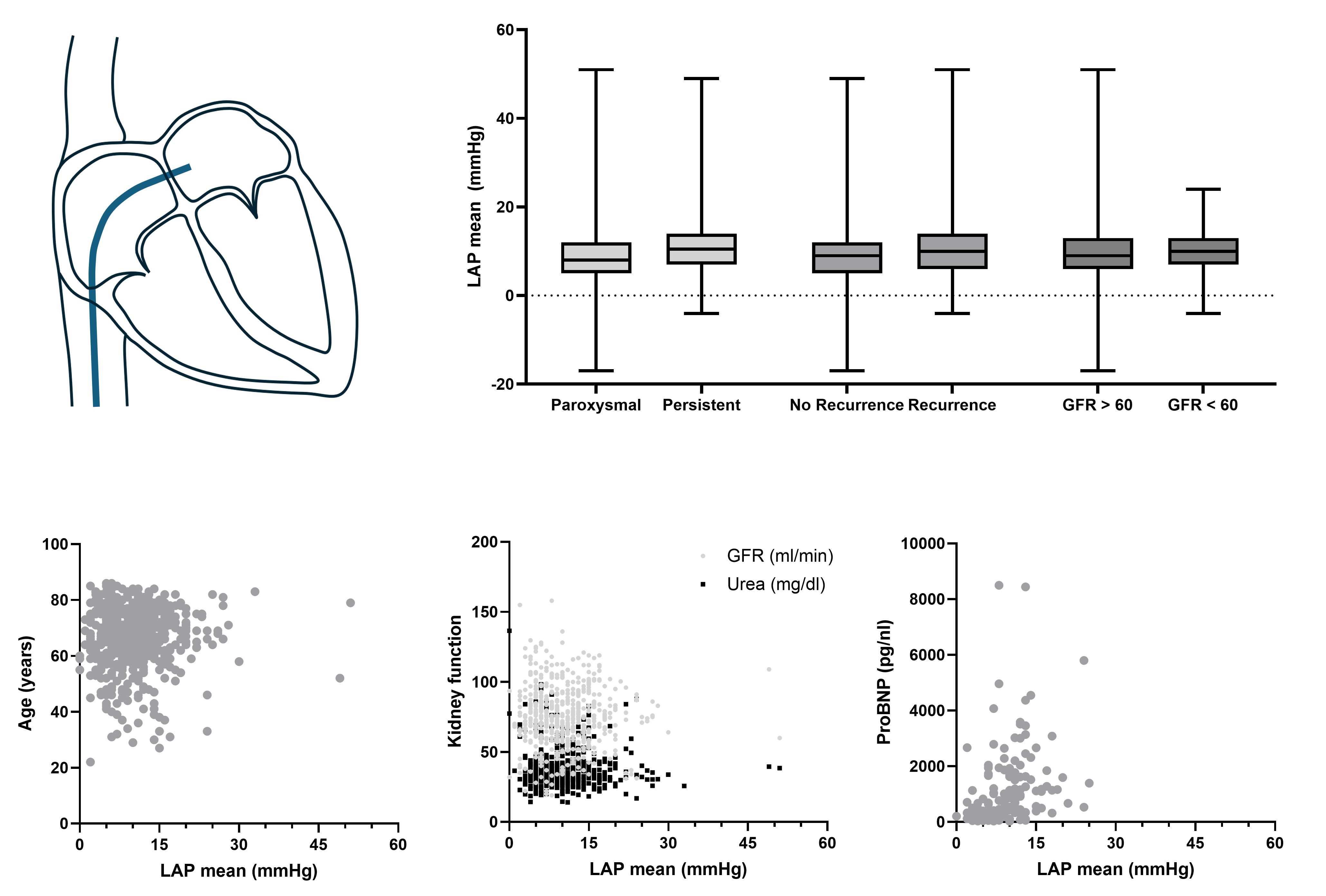
Results: We analyzed 577 patients (47% female; age 66.7±11.4 years; BMI 28.6±4.8 kg/m²; eGFR 75±21 ml/min/1.73m²; NT-proBNP 1087±1450 pg/mL; LVEF 55.7 ± 8.8%). AF was paroxysmal in 42% and persistent in 58%. Mean LAPm was 10.1±6.1 mmHg. 14% of patients had LAPm >15 mmHg. CKD stage ≥3 was present in 21%. Kidney function (eGFR) correlated negatively with age (ρ=−0.426), NT-proBNP (ρ=-0.377), and CHA₂DS₂-VASc (ρ=-0.348; all p<0.001), but not with AF recurrence (ρ=-0.077, p=0.067) or LAPm (ρ=-0.004, p=0.931). LAPm correlated with NT-proBNP (ρ= 0.467, p<0.001) and weakly with AF recurrence (ρ=0.141, p<0.001). In the ANCOVA, neither the interaction (F(1,559)=0.88, p=0.350) nor the main effects of recurrence (p=0.097) or eGFR (p=0.958) were significant (model R²=0.016; Levene p=0.533). In the multivariable logistic model (n=497), LAPm, eGFR, their interaction, and quadratic terms were not associated with AF recurrence (all p≥0.31). Female sex (OR 2.12, 95% CI 1.44–3.14, p<0.001), AF at TSP (OR 1.68, 95% CI 1.05–2.69, p=0.030), and persistent AF (OR 1.67, 95% CI 1.04–2.68, p=0.033) were independent predictors. The model had modest explanatory power (Nagelkerke R²=0.13) and good calibration (Hosmer p=0.758).
Conclusions: In first-time PVI, LAPm is strongly associated with NT-proBNP, but neither LAPm nor kidney function are linked to AF recurrence. Renal function does not moderate the relationship between LAPm and AF recurrence. Recurrence was more frequent in women, in patients with AF at TSP, and in persistent AF. These findings suggest that, in a predominantly non-severely CKD cohort, kidney function does not meaningfully influence LAPm or post-ablation AF recurrence.