Background Leaflet thrombosis following transcatheter tricuspid valve replacement (TTVR) is a rare but potentially serious complication that may compromise valve function and clinical outcome. With the increasing use of the Evoque system (Edwards Lifesciences, Irvine, CA, USA), evidence on optimal antithrombotic strategies remains limited, and standardized recommendations have yet to be established.
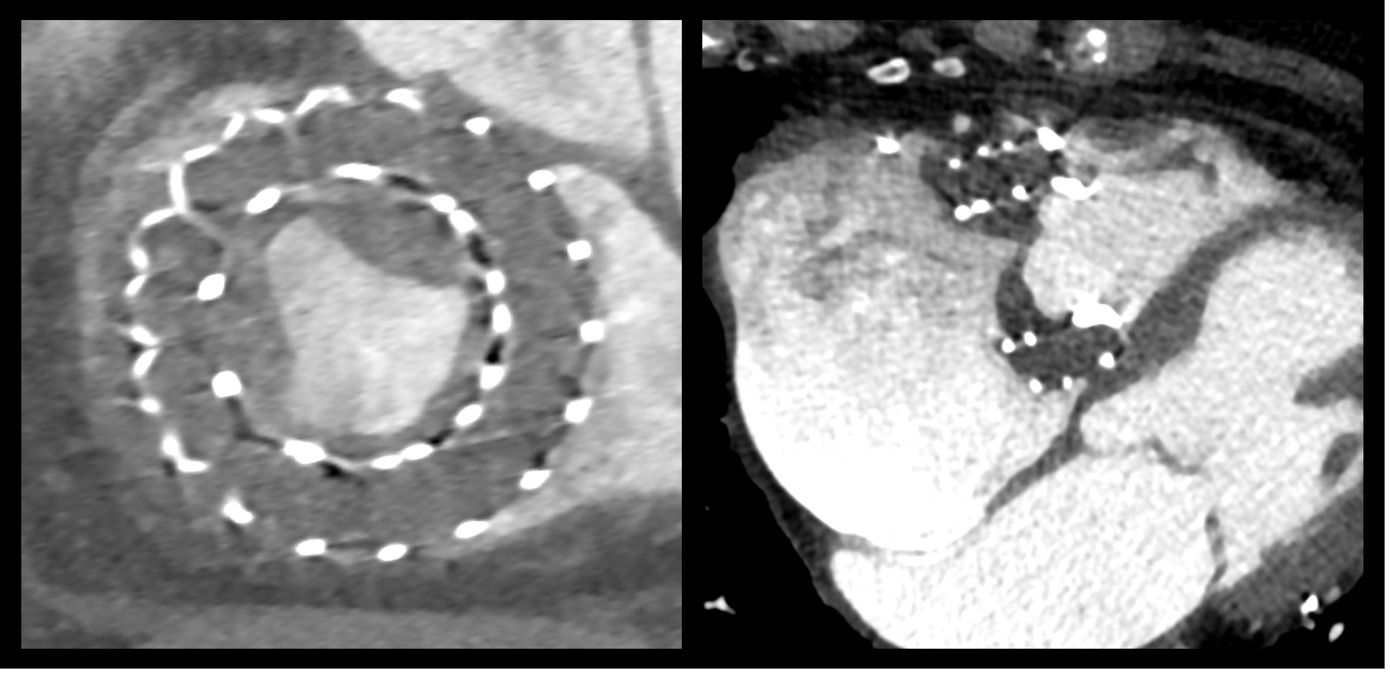
Case An 81-year-old woman with atrial fibrillation on apixaban, and a history of ischemic stroke presented with reduced exercise tolerance (NYHA III) and mild peripheral oedema. Echocardiography demonstrated preserved left ventricular systolic function (EF 60%), right atrial and right ventricular dilatation, and systolic flow reversal in the hepatic veins, consistent with torrential tricuspid regurgitation (EROA 0.9cm², regurgitant volume 60mL). She subsequently underwent TTVR with a 52 mm Evoque valve. The procedure was performed under general anaesthesia via right femoral venous access without complications. Regurgitation was completely eliminated with a resulting mean gradient of 1mmHg. Four days post-procedure, she developed complete atrioventricular block, requiring implantation of a single-chamber pacemaker using a bipolar coronary sinus lead. At 30 days, symptoms remained unchanged. Echocardiography showed a stable prosthesis with a mean gradient of 3.7mmHg. Cardiac CT revealed hypo-attenuated leaflet thickening (HALT) involving all three leaflets with reduced motion (RELM ≈ 50 %). Anticoagulation was switched from apixaban to phenprocoumon with a target INR of 2.0–3.0. Despite adequate anticoagulation, the 60-day follow-up showed further progression with complete leaflet immobility (RELM 100 %), an increased gradient (5.7mmHg), and deterioration of right-ventricular function (TAPSE 16 > 10 mm). The patient reported progressive dyspnoea. Low-dose systemic thrombolysis with alteplase (25mg over 25h) was administered under intensive care monitoring. No bleeding complications occurred. Post-lysis echocardiography revealed marked improvement with a mean gradient of 3.1 mmHg and partial recovery of leaflet motion. The patient was discharged in stable condition on phenprocoumon (target INR 2.5–3.0). Follow-up imaging is scheduled one month after discharge.
Conclusion This case illustrates a rare but clinically relevant complication after Evoque TTVR, successfully managed with low-dose systemic thrombolysis. It underscores the urgent need for standardized TTVR anticoagulation protocols. Moreover, the case exemplifies three complications increasingly reported after TTVR implantation - conduction disturbances, right-ventricular dysfunction, and valve thrombosis - emphasizing the importance of systematic surveillance and dedicated research to optimize long-term management.