Background: Despite remarkable therapeutic advances in the performance of transcatheter edge-to-edge mitral valve repair (mTEER), the occurrence of major adverse cardiovascular and cerebrovascular events (MACCEs) still remains a matter of debate. Owing to the continous increase in procedure numbers, the preliminary identification of subcohorts at elevated risk for MACCEs becomes increasingly relevant. However, current evidence on preprocedural predictors of MACCEs after mTEER is limited.
Methods: All patients who underwent mTEER in four german tertiary heart centers were investigated. A MACCE was defined as the occurrence of one or more of the following events during index hospitalization: Stroke or other thrombembolic event, bleeding according to the recommendations of the Mitral Valve Academic Research Consortium (MVARC), cardiac conduction system disturbances, myocardial infarction, and death during hospitalization. Primary endpoint of the study was survival after mTEER, which was compared via the Kaplan-Meier method between patients with and without MACCE during index hospitalization after adjustment of confounders via propensity-score-matching (PSM) in a 1:5 ratio. Predictors of MACCE were identified via uni- and multivariable logistic regression analyses.
Results: Of 821 patients who underwent mTEER, 5.48% (45/821) of cases were affected by MACCEs, which primarily consisted of minor bleeding complications (
Figure 1). Patients with occurred MACCEs had higher rates of coexistent comorbidites (
Table 1) and exhibited significantly poorer survival before and after PSM (
Figure 2). The multivariable logistic regression analysis identified euroSCORE II >15% (odds ratio [OR] 2.7, 95%-confidence interval [CI] 1.2-6.4, p=0.02), heart failure symptoms of New-York-Heart-Association (NYHA) class IV (OR 4.8, 95%-CI 2.4-9.6, p<0.001) and previous implantation of an implantable cardioverter defibrillator (ICD, OR 0.24, 95%-CI 0.07-0.6, p=0.01) as independent preprocedural predictors of MACCE during index hospitalization (
Table 2).
Conclusion: MACCEs during index-hospitalization were significantly associated with deteriorated survival, which was still evident after successful adjustment of confounders via PSM. The identified predictors can facilitate the early identification of patients at elevated risk for MACCEs and mortality.
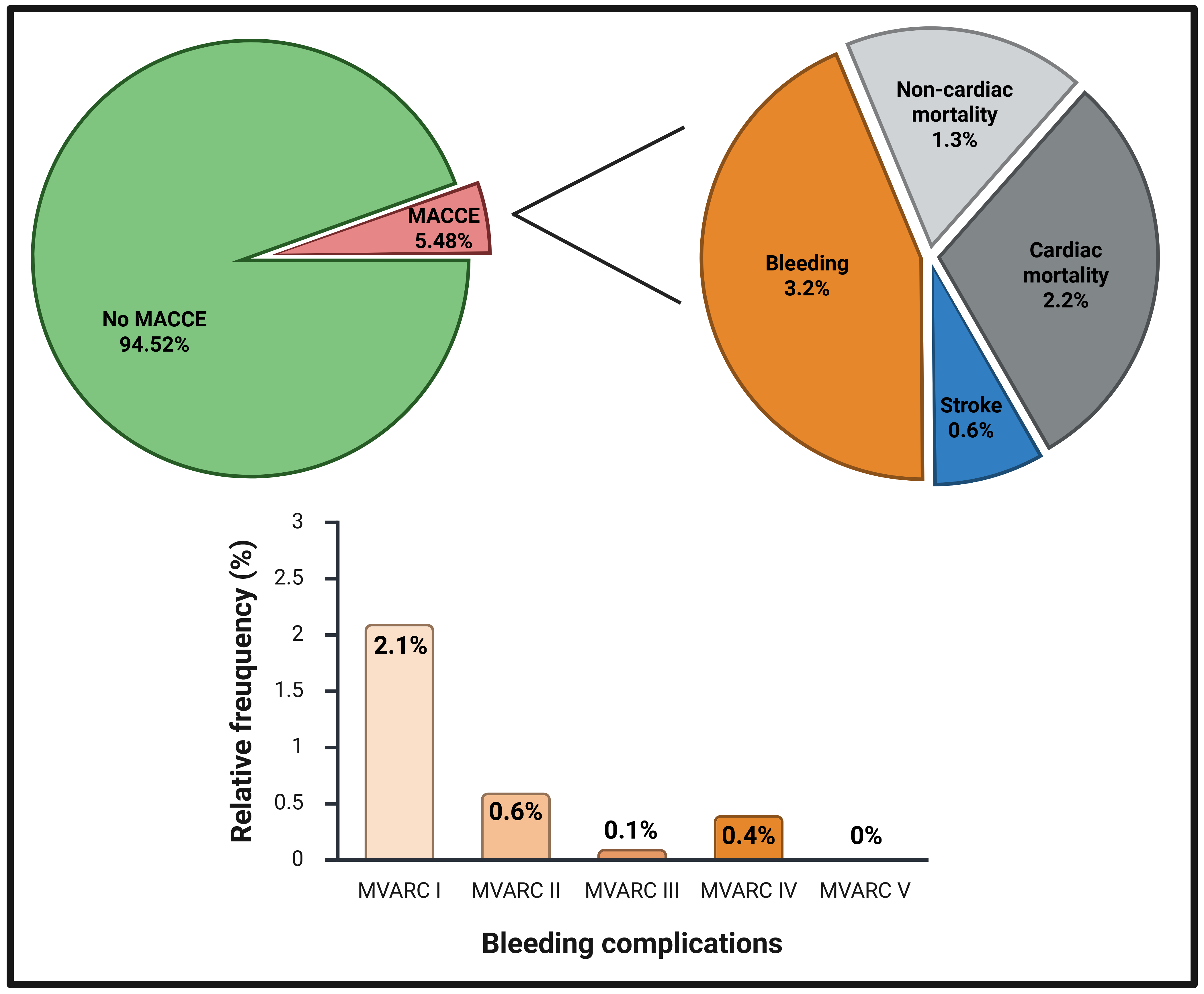 Figure 1: Distribution of MACCEs after mTEER
Figure 1: Distribution of MACCEs after mTEER
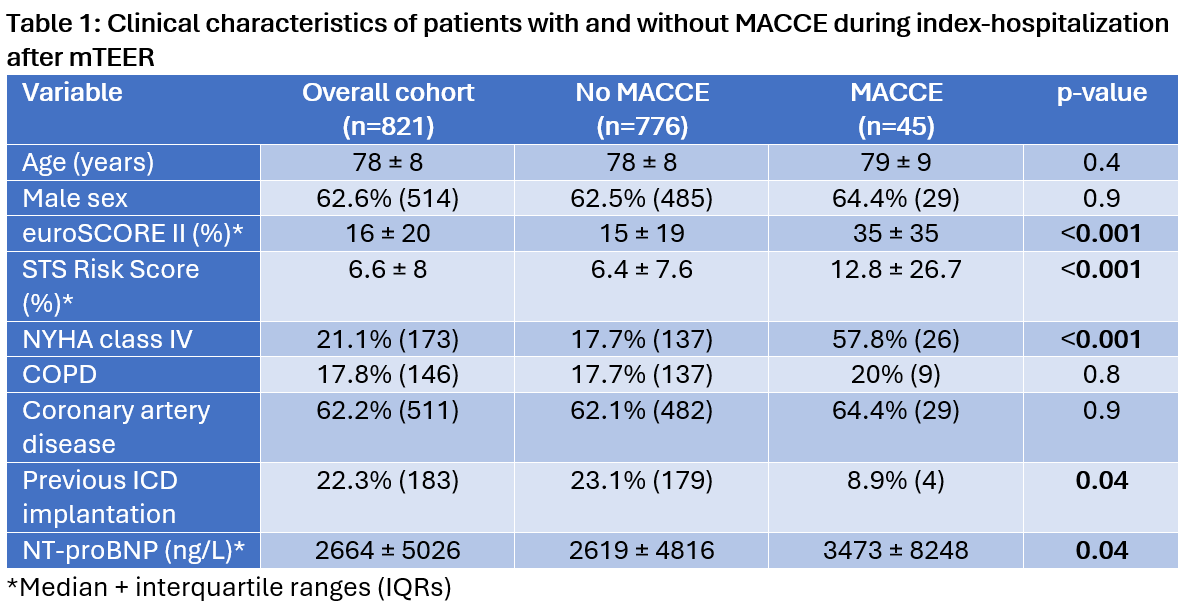
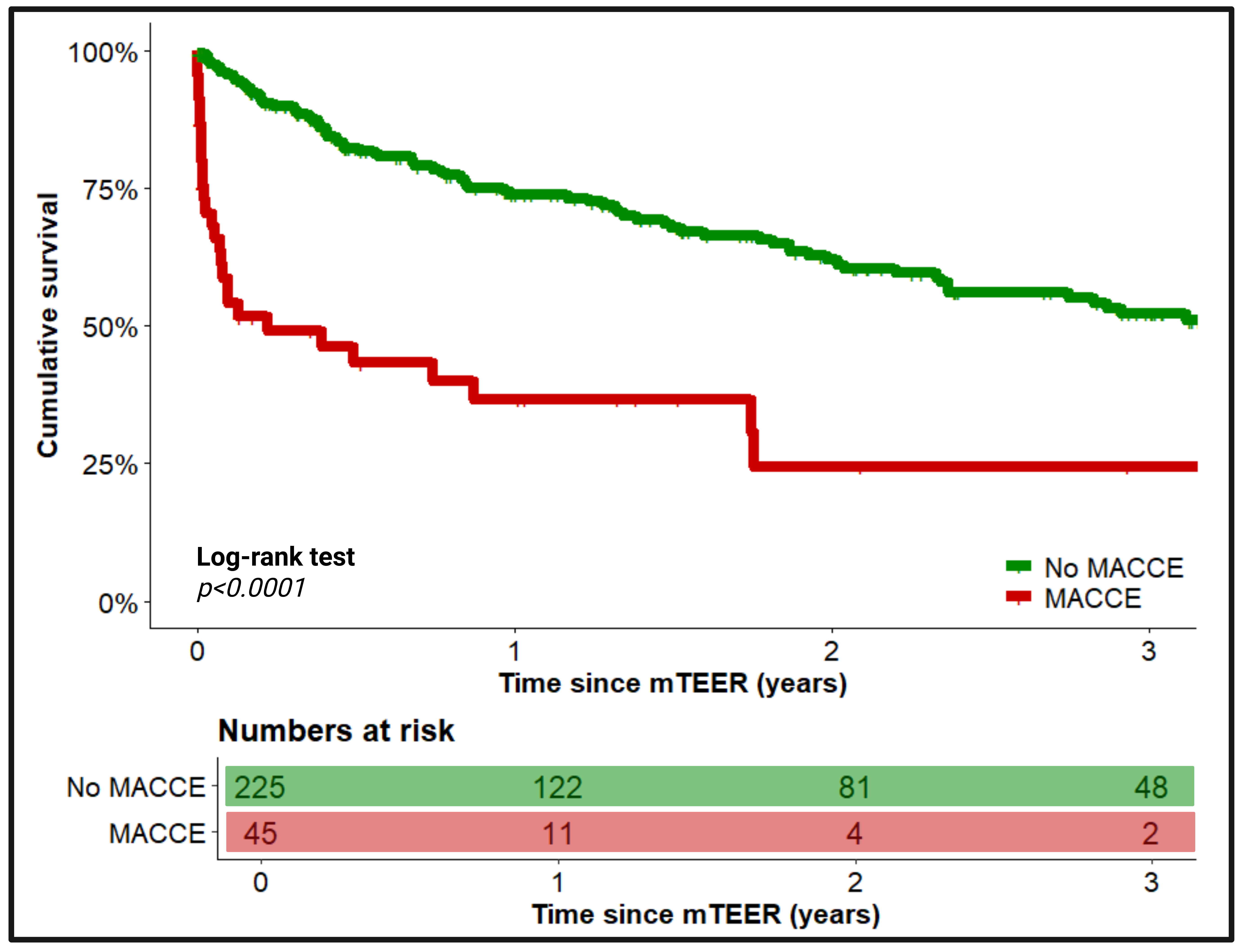
Figure 2: Long-term survival between patients with and without MACCE after mTEER after PSM

