Background:
Transcutaneous non-invasive vagal nerve stimulation is an emerging therapy in atrial fibrillation (AF), but selecting patients with appropriate cardiovagal responsiveness remains challenging.
Objective:
To assess whether baseline HRV or HRV changes during deep breathing are associated with cardiovagal responsiveness to auricular (aVNS) or cervical vagal nerve stimulation (cVNS).
Methods:
Patients with paroxysmal or persistent AF in sinus rhythm underwent standardized autonomic assessment. After a 5-minute rest, baseline HRV was recorded over 260 RR intervals. Three randomized interventions followed, each lasting 130 RR intervals: paced deep breathing (10-s cycles, 4:6 inspiration/expiration), aVNS, and cVNS using a transcutaneous stimulator (20 Hz, 200 ms, below pain threshold), separated by 5-minute washout phases. Relative changes (Δ%) in time-, frequency-, and non-linear HRV indices were analyzed. Appropriate cardiovagal response to aVNS and cVNS was defined as an increase in tonic (standard deviation of RR intervals, SDNN) and phasic vagal activity (root mean square of successive RR interval differences, RMSSD).
Results:
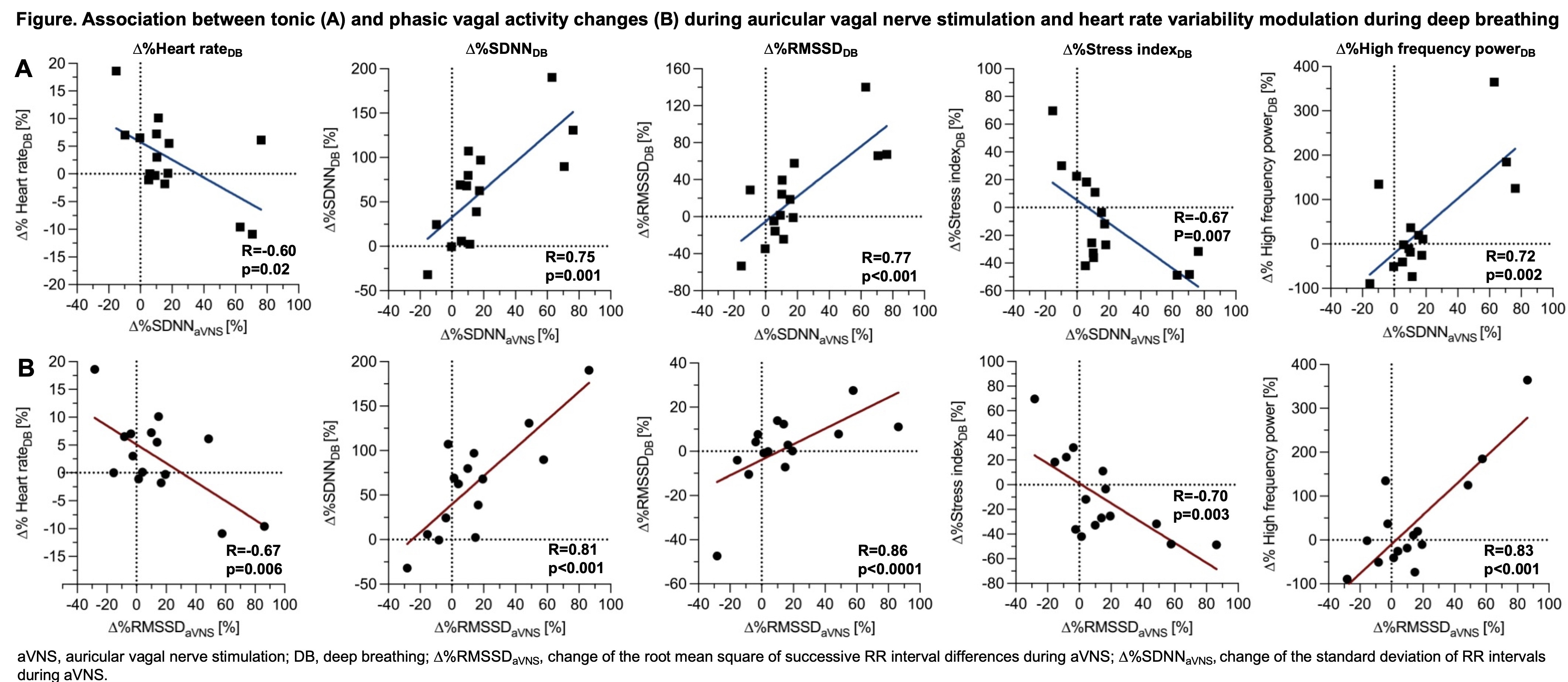
Fifteen patients were included (median age 67 [57; 75] years; 47% female; 87% paroxysmal AF; median CHA₂DS₂-VA 2 [0; 4]; 27% with prior AF ablation). Comorbidities included hypertension (53%), coronary artery disease (33%), diabetes (20%), obesity (20%), and heart failure (13%). Deep breathing increased tonic vagal activity (median SDNN: baseline 16 ms vs. deep breathing 27 ms vs. aVNS 19 ms vs. cVNS 22 ms; p < 0.001), while phasic vagal activity (median RMSSD: 19 vs. 20 vs. 19 vs. 24 ms; p = 0.54) and heart rate (median 59 bpm each; p = 0.12) remained unchanged. Appropriate cardiovagal response occurred in 67% during aVNS (median Δ%SDNN +16% and Δ%RMSSD +16%) and in 50% during cVNS (median Δ%SDNN +20%; Δ%RMSSD +39%). Appropriate responders did not differ in baseline vagal activity (SDNN 64 ms vs. 57 ms, p=0.37; RMSSD 17 ms vs. 26 ms, p=0.31) or heart rate response during deep breathing (expiratory/inspiratory difference 73 ms vs. 61 ms, p=0.79). In contrast, the magnitude of HRV change during deep breathing was strongly associated with tonic vagal activity changes during aVNS (∆%SDNNaVNS, Figure A), including Δ%SDNNDB (R = 0.81, p < 0.001), Δ%RMSSDDB (R = 0.77, p < 0.001), Δ%high-frequency powerDB (R = 0.72, p = 0.002), and Δ%SD1DB (R = 0.86, p < 0.001), with inverse associations for Δ%heart rateDB (R = –0.60, p = 0.02) and Δ%stress indexDB (R = –0.67, p = 0.007). Phasic responses showed similar associations (∆%RMSSDaVNS, Figure B). During cVNS, vagal changes correlated only with Δ%high-frequency powerDB during deep breathing (Δ%SDNNcVNS: R = 0.87, p = 0.001; Δ%RMSSDcVNS: R = 0.82, p = 0.004). No associations were observed with baseline HRV.
Conclusion:
In AF patients, autonomic changes during deep breathing are closely associated with cardiovagal responsiveness to non-invasive vagal nerve stimulation, whereas baseline HRV is not. Deep breathing may serve as a simple physiological tool to support patient selection for aVNS or cVNS.