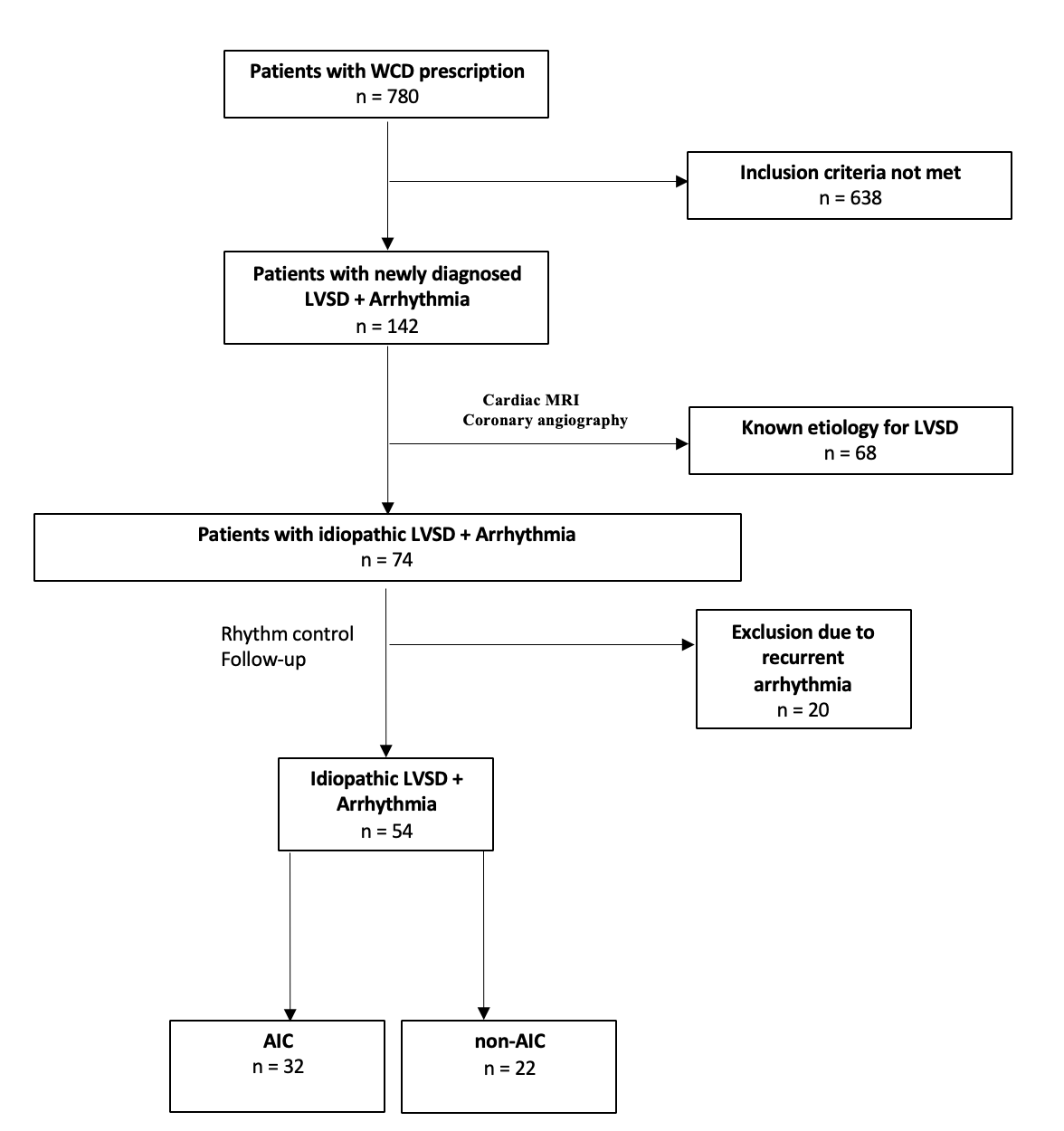
Background:
Arrhythmia-induced cardiomyopathy (AIC) is a potentially reversible cause of heart failure triggered by sustained arrhythmias. Its true real-world prevalence and clinical recognition remain limited.
Objective: This study aimed to assess the frequency, predictors, and outcomes of AIC in patients with newly diagnosed left ventricular systolic dysfunction (LVSD) and concurrent arrhythmia, undergoing rhythm control while protected by a wearable cardioverter-defibrillator (WCD).
Methods:
Among 780 WCD-treated patients (2017–2023), those with newly diagnosed idiopathic LVSD (LVEF < 35%) and concurrent arrhythmia (atrial fibrillation, flutter, or >20% ventricular ectopy) were analyzed. Diagnostic workup comprised echocardiography, cardiac MRI, and coronary angiography. Rhythm control was achieved via cardioversion, antiarrhythmic drugs, and/or ablation. Follow-up was up to 6 months. AIC was defined as LVEF improvement >15% with restored sinus rhythm.
Results:
Among 780 WCD patients, 142 (18.2%) had newly diagnosed LVSD in the presence of arrhythmia (Figure 1). Of these, 68 (47.9%) had an identifiable etiology: ischemic cardiomyopathy (n=38), valvular (n=10), myocarditis (n=12), sarcoidosis (n=2), Takotsubo (n=1), and amyloidosis (n=5). The remaining 74 (52.1%) with idiopathic LVSD were analyzed. At a mean follow-up of 138 days (4.5 months), 54 (73%) of 74 patients restored sinus rhythm, of whom 32 (59.3%) met AIC criteria. LVEF improved significantly in AIC patients (28% to 43%) compared to non-AIC patients (24% to 32%), with full recovery (>50%) in 56.3% of AIC patients. Atrial fibrillation was the most common arrhythmia. Heart failure therapy was comparable. Non-AIC patients exhibited larger left ventricular end-diastolic (LVEDD) and end-systolic diameters (LVESD), lower presenting heart rates, more mitral regurgitation. Non-AIC patients showed a trend toward more frequent late gadolinium enhancement (LGE) on cardiac MRI (p = 0.073). However, none of these parameters reliably predicted the presence of AIC. LVEF < 25% predicted lack of full recovery (p = 0.04). No appropriate WCD shocks occurred. ICD indication dropped from 100% to 17% (9/54) after rhythm control and optimized heart failure therapy in AIC and Non-AIC patients (36.4% versus 3.1%, p = 0.001).
Conclusions:
AIC is a common and reversible form of LVSD in patients with arrhythmia and newly diagnosed idiopathic cardiomyopathy. Early rhythm control supported by WCD, combined with detailed diagnostics, enables the identification of reversible LVSD and may reduce unnecessary ICD implantation.