Background. Myocardial fibrosis (MF) is a key determinant of adverse outcomes in severe aortic valve stenosis (AS) and limits the efficacy of valve replacement. Endomyocardial biopsy remains the reference standard for MF identification, but its invasiveness restricts routine use. Cardiovascular magnetic resonance (CMR)–derived extracellular volume (ECV) offers a noninvasive surrogate, yet correlates only modestly with biopsy-proven MF. Comprehensive data extraction through CMR, as reflected by radiomics, may improve noninvasive MF detection. We therefore investigated whether radiomics analysis from routine CMR enhances identification of increased biopsy-proven MF burden in severe AS.
Methods. Between 2017 and 2022, we prospectively enrolled 56 patients with severe AS scheduled for transcatheter aortic valve replacement (TAVR) at the University Medical Center Göttingen. All patients underwent pre-procedural 3T CMR and intraprocedural endomyocardial biopsy. Histologic MF was quantified by digital morphometry (Masson’s trichrome), classifying patients as high (≥11% collagen area fraction) or low fibrotic burden per published criteria. CMR mid-ventricular short-axis myocardium was manually segmented on cine images using an in-house-built Python tool. Radiomics features (n=1,023) were extracted using the PyRadiomics package, encompassing first-order statistics and texture metrics. Only features with a standard deviation >0.10 and intra-/interobserver correlation ≥0.80 were included, then reduced to five principal components (PCs) explaining >85% of variance. A logistic regression analysis with five-fold cross-validation was used to model the association between radiomics PCs and MF. Model performance was evaluated using the area under the receiver operating characteristic curve (AUC) and net reclassification improvement (NRI), compared with an ECV-only model and a combined ECV + radiomics PCs model.
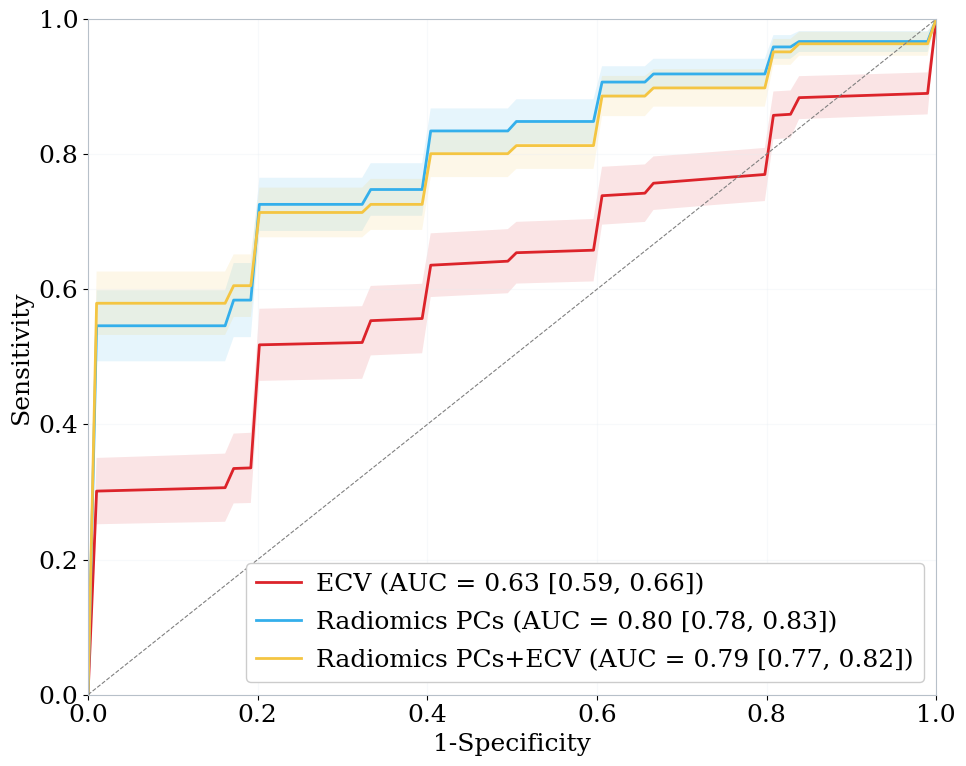
Results. Median age was 80 [77–83] years, and 30% were female. A total of 29 patients (52%) had a high MF burden. There were no differences in demographic characteristics, comorbidities, or echocardiographic parameters between patients with high and low MF burden (all p>0.05). Patients with higher MF had larger LV end-diastolic volumes (92 vs 76mL/m²; p=0.018) and showed a trend towards higher ECV (28% vs 26%; p=0.072). For detecting an increased myocardial fibrosis burden, the ECV-only model achieved an AUC of 0.63 (95% CI: 0.59–0.66). A model based on five radiomics PCs outperformed ECV alone (AUC 0.80 [0.78–0.83]). Adding ECV to the radiomics model did not provide additional discriminatory benefit (AUC 0.79 [0.77–0.82]; Figure 1). The radiomics model significantly improved noninvasive discrimination, with an NRI of 0.66 [0.59–0.73], NRI positives=0.34, and NRI negatives=0.32.
Conclusion. CMR-derived radiomics improved noninvasive detection of biopsy-proven myocardial fibrosis in severe aortic stenosis, supporting its potential role in precision phenotyping and personalized management.