Background: Exosomes are nano-sized small extracellular vesicles that are secreted by virtually all cell types, including cardiomyocytes, under both homeostatic and diseased conditions. They carry cell-specific proteins, lipids, and nucleic acids that reflect the molecular and functional state of their cell of origin and mediate intercellular communication. Their lipid bilayer protects exosomal cargo from enzymatic degradation, ensuring stability in circulation and making them attractive candidates for biomarker discovery.
Objective: We aim to investigate whether cardiomyocytes release exosome-bound cardiac troponin under hypoxic conditions, and to quantify circulating exosomes in a murine model of myocardial infarction induced by left anterior descending (LAD) coronary artery ligation, as well as in patients with myocardial injury.
Methods: Rat H9c2 cardiomyoblasts were differentiated into cardiomyocyte-like cells using all-trans retinoic acid, resulting in increased cellular cTnT expression, as described previously (Branco et al., PLoS ONE 2015). Differentiated cells were exposed to hypoxia (1.5% O₂) for 6 and 24 hours respectively, and exosomes were isolated to assess cTnT enrichment. To examine whether myocardial injury is associated with elevated circulating exosomes at the organismal level, we employed a murine model of myocardial infarction induced by minimally invasive left anterior descending (LAD) artery ligation (Sicklinger et al., Circ Res 2020). Serum exosome concentrations were measured 4 hours post-infarction using nanoparticle tracking analysis. To assess clinical relevance, serum samples from patients with non-ST elevation myocardial infarction (NSTEMI) and healthy controls were analyzed for serum exosome concentration and exosome-associated cTnT using immunoprecipitation with a high-sensitive cTnT capture antibody.
Results: In vitro, differentiated H9c2 cells exposed to hypoxia exhibited increased cellular cTnT expression and enrichment of cTnT in secreted exosomes. In mice, LAD ligation induced anterior wall infarction, resulting in elevated serum free cTnT levels, and an approximately three-fold increase in circulating exosome concentration compared to sham controls, without significant changes in exosome size. In NSTEMI patients, serum exosome concentrations were similarly increased (~three-fold) compared to healthy controls, while average exosome size remained unchanged. Immunoprecipitation assays confirmed enrichment of cTnT within exosomal isolates in NSTEMI patients.
Conclusion: Cardiomyocytes release exosome-bound cTnT under hypoxic stress in vitro. Myocardial hypoxia induces a systemic increase in circulating serum exosomes in both murine models and NSTEMI patients. These results support the concept that exosome-associated cardiac troponin may serve as an early biomarker of myocardial injury.
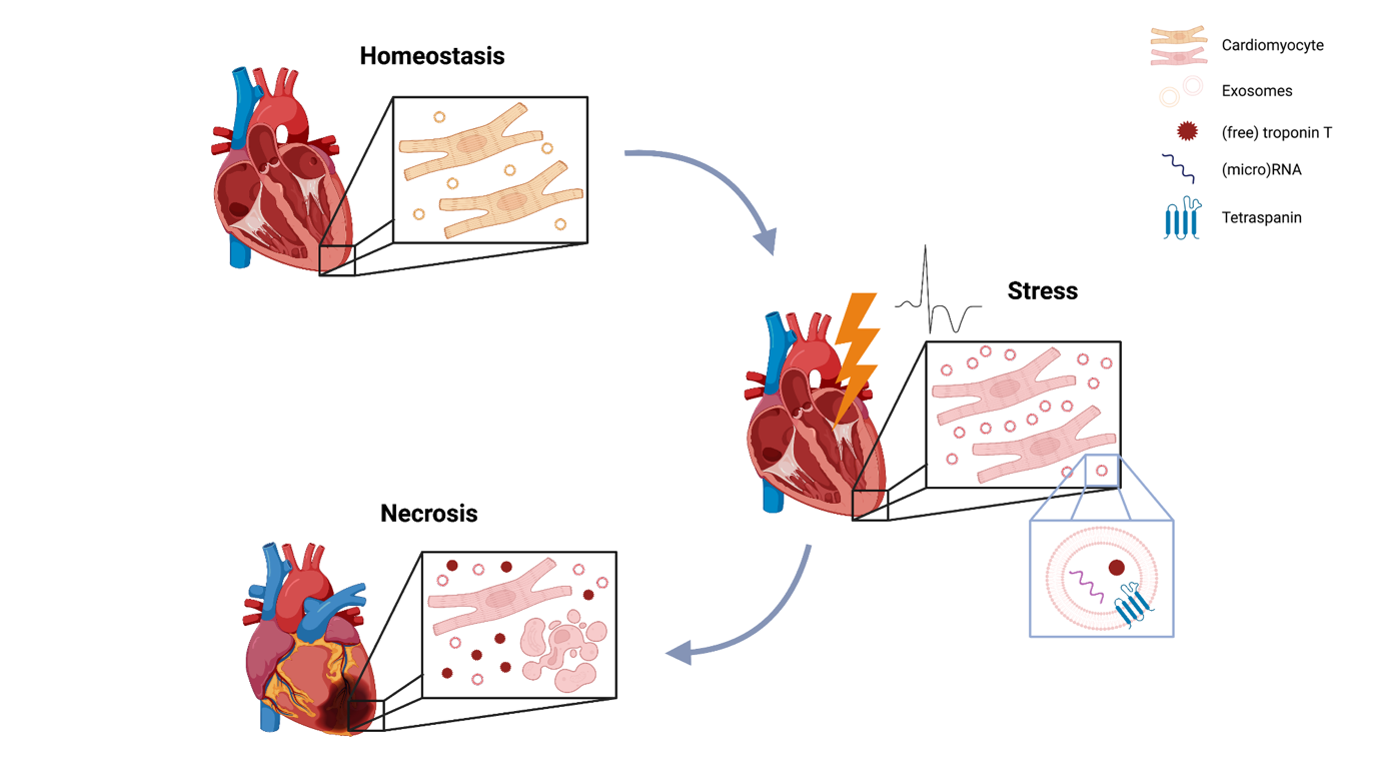
Fig. 1: Exosomal cardiac troponins, which have so far not been considered in routine clinical diagnostics, have the potential to enable early detection of myocardial disease, as they are released under early hypoxic, pre-necrotic conditions.
