Background: Anthracyclines represent a cornerstone in contemporary anti-cancer therapy, but are limited by dose-dependent, often irreversible cardiotoxicity. The molecular mechanisms underpinning anthracycline-induced cardiotoxicity (AIC) remain poorly defined, impeding the development of targeted cardioprotective strategies. This study aimed to integrate high-resolution myocardial proteomics with metabolic network modeling to identify key molecular mechanisms and therapeutic targets in anthracycline-induced cardiotoxicity.
Methods: Left ventricular biopsies were obtained from patients with advanced AIC (n=17; 53±8 years) and age-matched non-failing donor hearts (n=15). Myocardial proteins were quantified using high-resolution label-free mass spectrometry. Differentially expressed proteins were functionally annotated through Gene Ontology (GO) enrichment analyses. Based on the individual proteomics-derived enzyme abundance profiles, patient-specific computational models of myocardial metabolism were generated, encompassing 296 biochemical reactions across all main energy-generating pathways. Proteomic and metabolic alterations were correlated with clinical characteristics, cardiac imaging parameters, and outcomes.
Results: Patients with AIC exhibited severely reduced left ventricular systolic function (LVEF 20±5%) and high symptom burden (76.5% NYHA class IV) despite guideline-directed medical therapy. Most had received anthracyclines as part of combination chemotherapy for non-Hodgkin lymphoma or breast cancer (76.7%). Among 6,940 identified proteins, 527 (7.6%) were significantly upregulated and 305 (4.4%) downregulated in AIC. Upregulated proteins were predominantly involved in pathways related to inflammation and adaptive immune activation, whereas downregulated proteins were enriched in mitochondrial respiration and ATP synthesis (FDR for all <0.01). Personalized reconstruction of myocardial metabolism revealed a substantially reduced maximal ATP production capacity, a shift in myocardial substrate utilization from fatty acids towards glucose oxidation, and impaired uptake capacities for ketone bodies and branched-chain amino acids, delineating a profound bioenergetic derangement in AIC.
Conclusions: Large-scale myocardial proteomics revealed disease-specific molecular alterations in anthracycline-induced cardiotoxicity, providing new mechanistic insights into metabolic remodeling and identifying potential molecular targets for cardioprotection in cancer survivors.
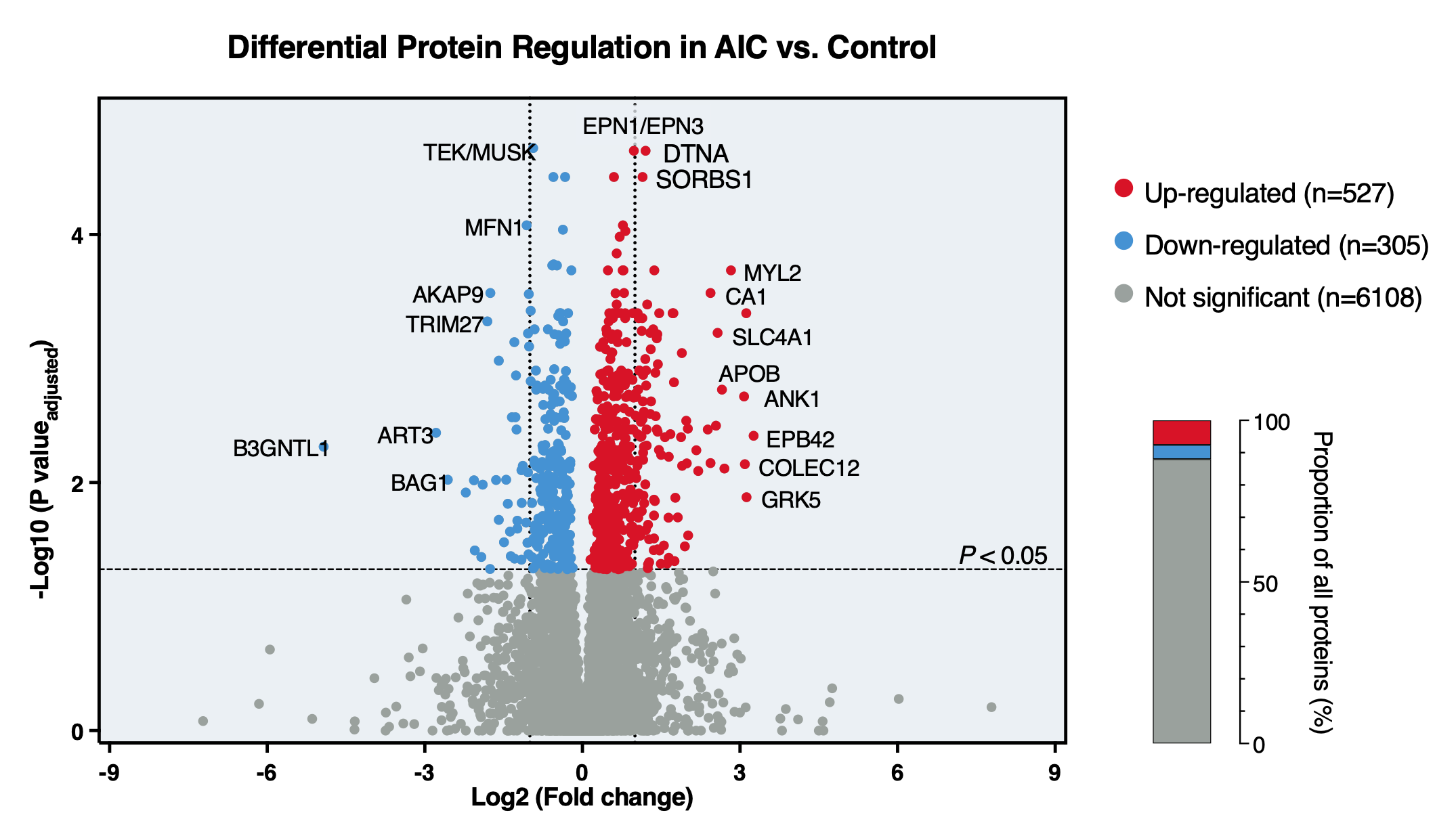
Figure 1: Differential protein expression analysis of myocardial biopsies obtained from patients with AIC and non-failing controls.
