Background: Acute hemodynamic decompensation (AHD) remains a serious periprocedural complication during catheter ablation for ventricular tachycardia (VT). Rescue mechanical circulatory support (MCS) is often required in these cases but is associated with poor outcomes. The PAINESD and PAAINESD scores have been proposed to stratify patients at increased risk for periprocedural AHD and to guide prophylactic MCS use. However, independent validation in large, contemporary VT ablation cohorts is limited.
Aims: This study aimed to (1) validate the PAINESD and PAAINESD scores in a large single-center cohort of VT ablation patients, (2) compare periprocedural outcomes between low- and high-risk groups for AHD, and (3) identify independent predictors of AHD.
Methods: This study retrospectively analyzed consecutive patients undergoing VT ablation between 2018 and 2025. The primary endpoint was periprocedural AHD, defined as persistent cardiogenic shock despite vasopressor therapy. Secondary outcomes included in-hospital mortality, intensive care unit (ICU) admission, mechanical ventilation, and major procedural complications (pericardial tamponade, major bleeding, thromboembolic events). Patients were categorized as low-risk (PAINESD <15 and PAAINESD ≤17) or high-risk (PAINESD ≥15 or PAAINESD >17). Logistic regression models were applied to identify independent predictors for AHD.
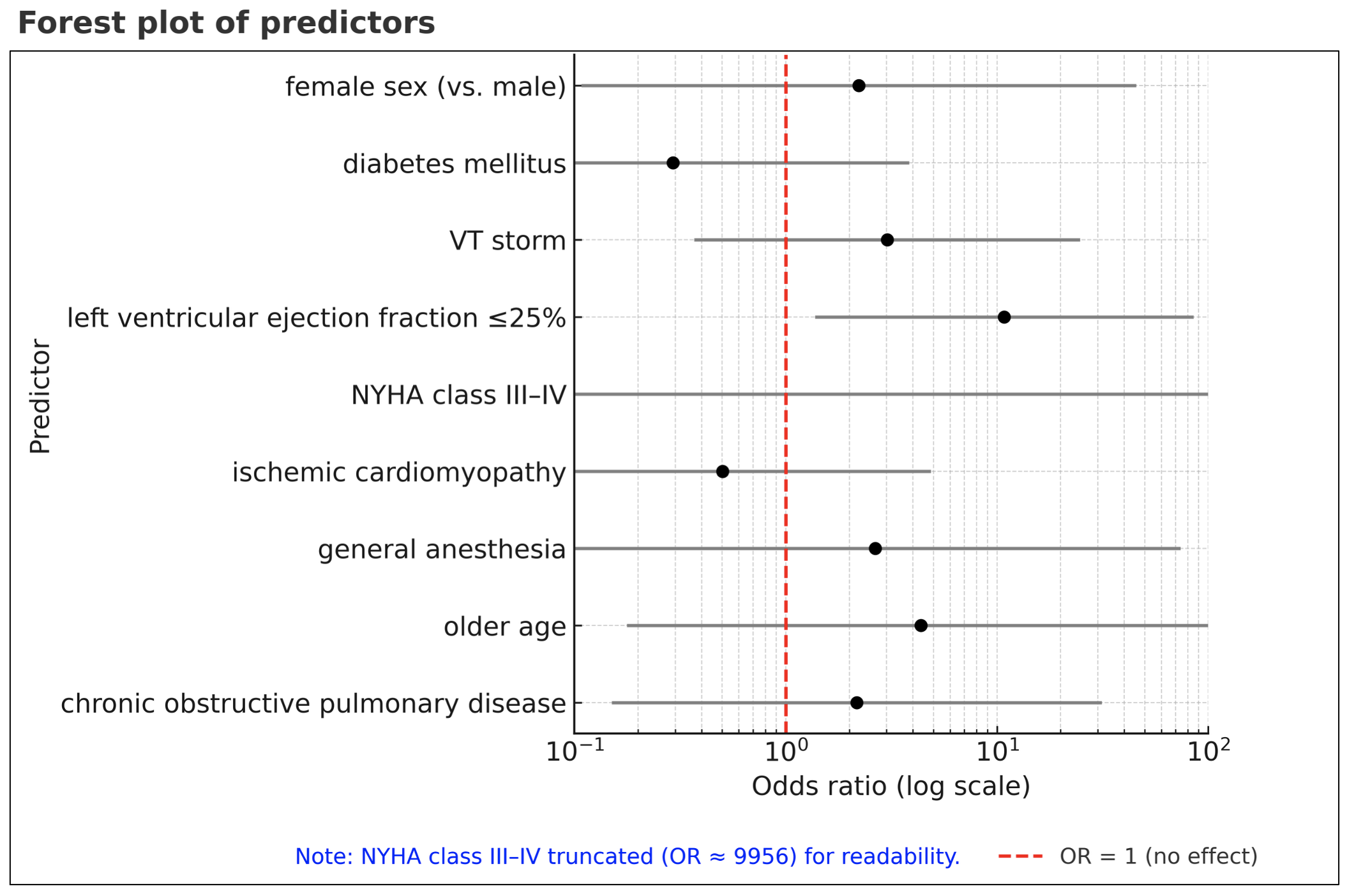
Results: A total of 339 patients (mean age 66 ± 11 years; 12% female; 56% ischemic cardiomyopathy, 29% non-ischemic cardiomyopathy) were included. The mean PAINESD and PAAINESD scores were both 9 ± 6. The primary outcome occurred in six patients (1.8%), including two requiring percutaneous ventricular assist devices (0.6%), three extracorporeal membrane oxygenation (0.9%), and two levosimendan laoding (0.6%). In-hospital mortality was 2.1%. AHD was significantly more frequent in the high-risk group (6.0%) compared with the low-risk group (0.7%). High-risk patients also showed increased rates of in-hospital mortality (7.5% vs. 0.7%), ICU admission (24% vs. 8.5%), and mechanical ventilation (12% vs. 2.6%) following ablation, whereas rates of pericardial tamponade, major bleeding, and thromboembolic events were similar between groups. In multivariable analysis, high-risk classification was associated with higher odds of AHD (OR 8.6, 95% CI 1.5–48.1, p=0.014). Among individual score components, a left ventricular ejection fraction <25% was the only independent predictor for AHD (OR 10.8, 95% CI 1.4–85.3, p=0.024).
Conclusion: The PAINESD and PAAINESD scores effectively identify VT ablation patients at elevated risk for periprocedural AHD. However, among individual clinical variables, only severely reduced left ventricular ejection fraction (<25%) independently predicted AHD. These findings support targeted risk stratification and selective prophylactic MCS use in high-risk patients.