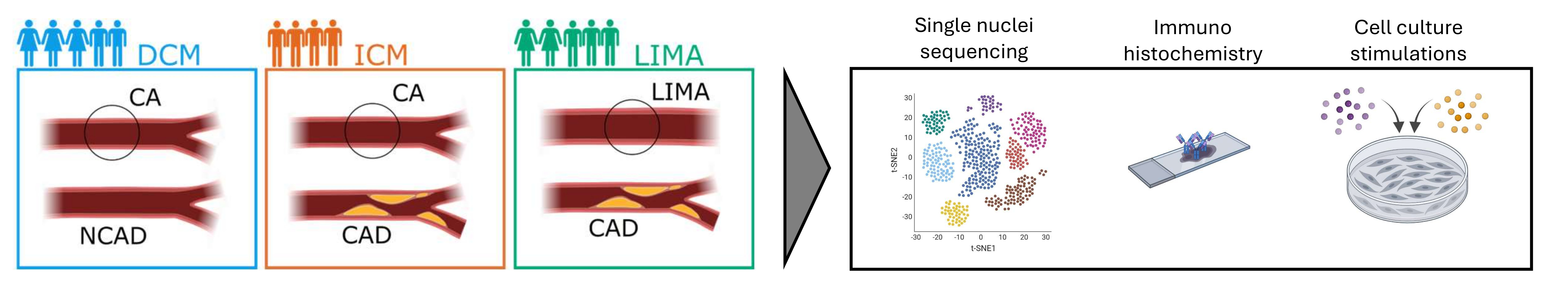
Background:
Clinical observations from patients undergoing coronary artery bypass grafting have shown that the internal mammary artery (LIMA) exhibits remarkable resistance to atherosclerosis compared to coronary arteries (CA), despite similar exposure to systemic risk factors. The molecular and cellular mechanisms underlying this atheroresistance remain poorly understood.
Objectives and Experimental Design:
To elucidate the underlying mechanisms, we performed single-nucleus RNA sequencing (snRNA-seq) on non-atherosclerotic CAs from patients without coronary artery disease (DCM) and with CAD (ICM) undergoing heart transplantation, as well as on LIMA samples from CAD patients undergoing bypass surgery. Unsupervised clustering of all nuclei was performed, and cell types were identified using canonical marker genes. Cell-cell interaction probabilities were inferred from ligand-receptor expression profiles. Additionally, snRNA-seq findings were validated on tissue sections, and endothelial cell (EC) culture stimulations were performed to explore gene expression responses.
Results:
Unsupervised clustering identified ten major cell types, including two smooth muscle cell (SMC) populations, ECs, fibroblasts, myeloid and lymphoid cells, glial cells, and adipocytes, which are identified using canonical marker genes. All cell types were present across the three groups, although lymphoid (1.3% DCM, 2.1% IC, 0.2% LIMA), myeloid (3.8% DCM, 4.2% ICM, 1.5% LIMA) and adipocyte (0.5% DCM, 0.6% ICM, 0.03% LIMA) fractions were significantly lower in LIMA compared to CA samples.
SMCs were the predominant cell population in all samples (~64% in DCM, ~71% in ICM, ~70% in LIMA). Ligand-receptor analysis revealed distinct signalling profiles: IGF1-IGF1R, BMP6-BMPR1A/BMPR2, and CD46-JAG1 interactions were predicted in ICM but absent in LIMA. Subclustering of SMCs uncovered five transcriptionally distinct subclusters (SMC1-5), with SMC1 enriched in CA and SMC2 in LIMA, consistent with vessel-specific SMC heterogeneity previously described between the pulmonary artery and aorta.
ECs accounted for 9%, 6%, and 4% of total nuclei in DCM, ICM, and LIMA, respectively. Given the reduced immune cell presence in LIMA, we focused on altered EC-immune cell interactions across groups. CD6-ALCAM signalling was exclusive to ICM, in line with elevated ALCAM expression in ICM-derived ECs. In contrast, CD99-related interactions were absent in LIMA, reflecting lower CD99 expression in LIMA-ECs – a finding confirmed histologically. Furthermore, pro-inflammatory stimulation of cultured ECs (HMECs) upregulated both ALCAM and CD99 expression, while stimulation with oxidized Low-Density Lipoprotein only upregulated CD99 but not ALCAM expression, supporting context-dependent regulation of these molecules.
Conclusion:
Our data reveal distinct cellular compositions, transcriptomic and signalling landscapes between CA and LIMA. The absence of pro-inflammatory ligand–receptor interactions in LIMA-ECs may contribute to its atheroresistant phenotype. These findings highlight cell-type-specific molecular differences that could inform strategies for vascular protection and graft longevity in CAD.