Background:
The “leave-nothing-behind” strategy aims to overcome long-term limitations of drug-eluting stents (DES) by avoiding permanent vessel caging and late thrombotic risk. The bioresorbable magnesium scaffold Magmaris™ gained CE-mark in 2016, offers transient vascular support and gradually dissolves as healing progresses. In acute coronary syndrome (ACS), plaque rupture usually involves non-calcified lesions with minimal fibrosis and temporary scaffolding may be sufficient for these lesions. With the next-generation magnesium scaffold FREESOLVE™ now in clinical evaluation, consolidating the evidence from Magmaris™ is essential to define the safety and efficacy of magnesium-based bioresorbable scaffolds in ACS.
Aims:
To evaluate safety and efficacy of Magmaris™ in ACS and compare outcomes with contemporary DES.
Methods:
PubMed, Embase (Ovid), and the Cochrane Central Register (Ovid) were searched for randomized and observational studies investigating Magmaris™ in ACS. Outcomes were target-lesion failure (TLF), target-lesion revascularization (TLR), and scaffold thrombosis (ST) at 1-year follow-up. Pooled event rates were calculated using generalized linear mixed-effects models (GLMM). Pairwise analyses between Magmaris™ and DES applied random-effects GLMMs to estimate odds ratios (ORs) with 95%CIs. Heterogeneity was assessed using I² and Wald tests; p<0.05 was considered significant.
Results:
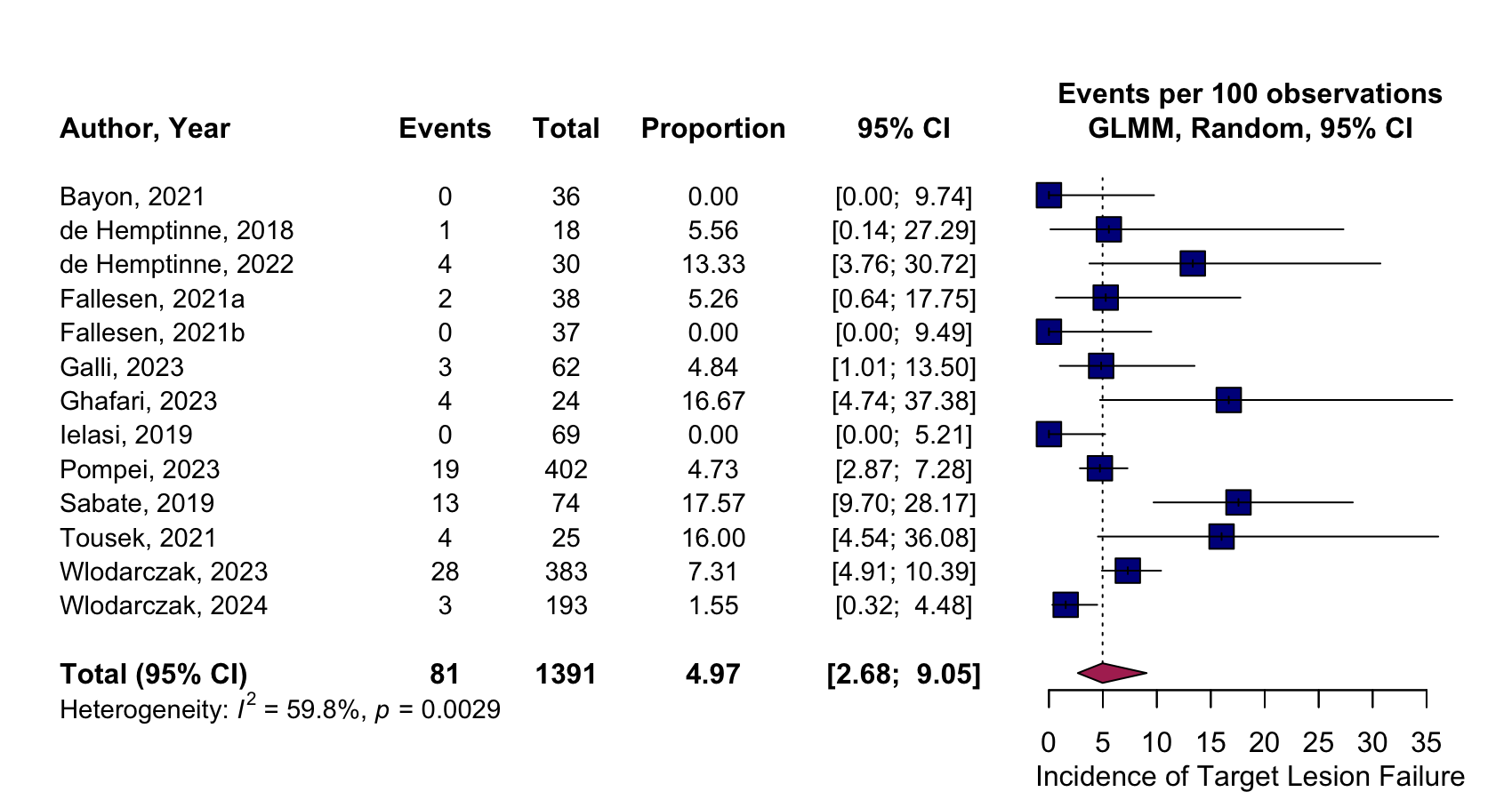
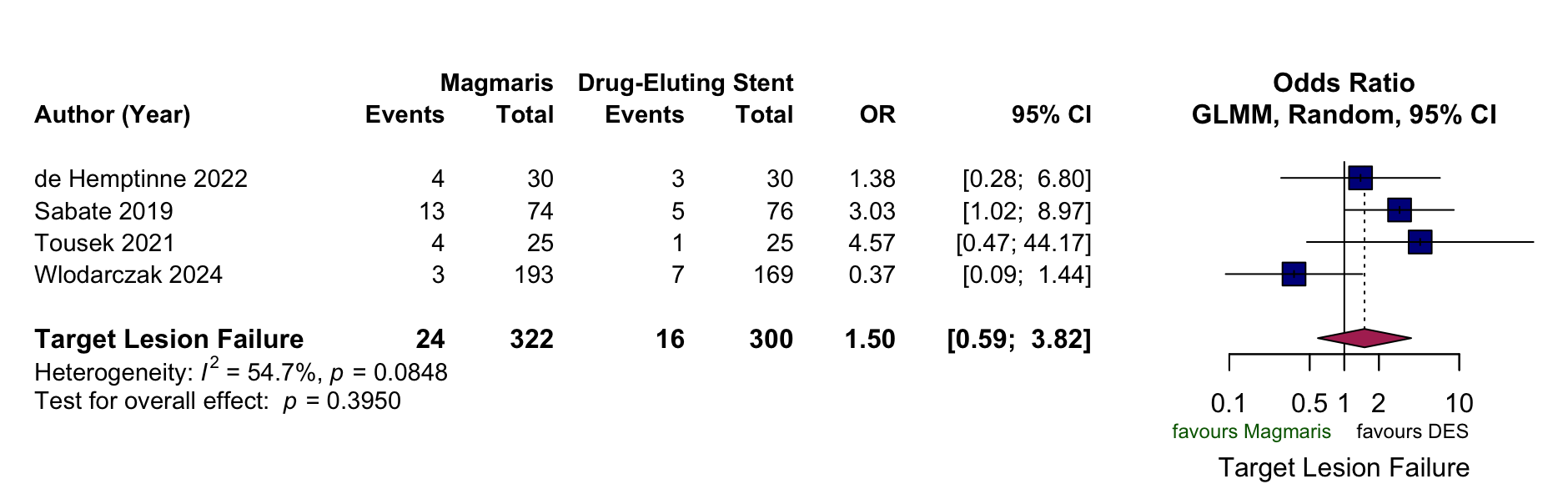
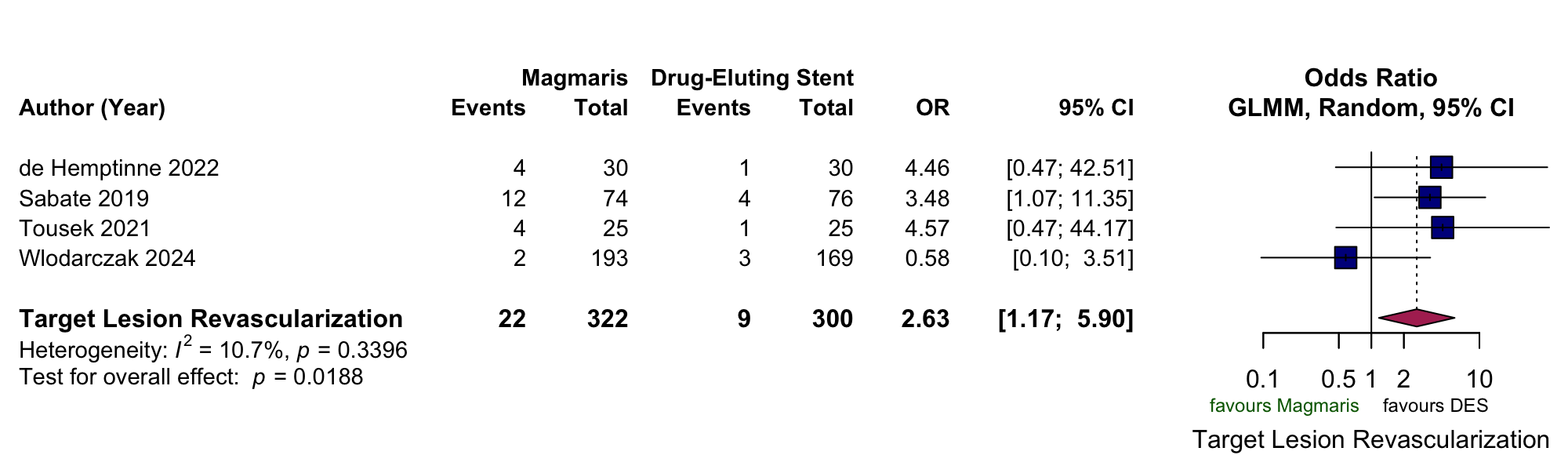
Four studies (622 patients in total; 322 Magmaris™, 300 DES) were included in the pairwise analysis. Mean age was 62.7 ± 10.6 (Magmaris™) and 62.1 ± 10.1 years (DES). Clinical presentation included 122 STEMI (37.9%) and 200 NSTEMI (62.1%) for Magmaris™, and 120 STEMI (40.0%) and 180 NSTEMI (60.0%) for DES. 12 studies (1391 patients) were included in the single-arm analysis. Mean age was 58.5 ± 10.5 years; clinical presentation 376 STEMI (27.0%), 946 NSTEMI (68.0%), 69 UA (5.0%). At 1 year, TLF was 7.5% vs 5.3% (OR 1.50, 95%CI 0.59–3.82; p=0.39; I²=54.7%), TLR 6.8% vs 3.0% (OR 2.63, 95%CI 1.17–5.90; p=0.019; I²=10.7%), and ST 0.6% vs 1.0% (OR 0.67, 95%CI 0.11–4.09; p=0.67; I²=44.8%) for the Magmaris and DES arm, respectively . In the single-arm analyses, TLF was 4.97% (95%CI 2.68–9.05; I²=59.8%), TLR 5.03% (95%CI 4.00–6.31; I²=57.8%) and ST 0.83% (95%CI 0.43–1.60; I²=28.3%). Procedural success was 98.49% (95%CI 97.21–99.18; I²=35.4%).
Conclusion
Accounting for all existing evidence in literature, Magmaris™ shows high procedural success and low scaffold thrombosis rates with overall safety comparable to DES, while higher TLR rates highlight the need for further refinement of fully bioresorbable technologies in ACS.
Figure 1: Pooled outcomes of selected pairwise and single-arm meta-analyses. A: Target-lesion failure at one-year: Magmaris™ vs. DES. B: Target-lesion revascularization at one-year: Magmaris™ vs DES. C: Target-lesion failure at one year: Magmaris™.
A
B
C