Background and aims: Atherosclerosis and its complications remain a leading cause of morbidity and mortality around the globe. In the context of atherosclerosis, peripheral blood mononuclear cells (PBMCs) exhibit a phenotypic spectrum depending on the local micromilieu. A distinct subtype of PBMC arises in the presence of CXCL4 (platelet factor 4) which is secreted by activated platelets. These CXCL4-polarized PBMCs have pro-inflammatory and pro-atherogenic effects and low phagocytic capacity. CXCL4-dependent differentiation of PBMCs also leads to an enhanced secretion of extracellular vesicles (EV) that induce a pro-calcific, pro-inflammatory state in adjacent vascular smooth muscle cells. To gain deeper mechanistic insights into the contribution of these EV in atherogenesis, we conducted a vesiculomics study to analyze their miRNA and protein cargo and investigated their spatial distribution in ex-vivo murine carotid artery plaques.
Methods: PBMCs were isolated from the blood of healthy human donors and cultured for seven days. PBMCs were differentiated with either CXCL4 or M-CSF (macrophage colony stimulating factor) as control. EV were isolated from cell culture supernatant and prepared for further analysis. Bulk RNA-Sequencing was performed to identify differentially enriched vesicular miRNA transcripts. The upregulation of miRNA of interest in the context of atherosclerosis was confirmed via qRT-PCR. Following unbiased proteomics analysis, differentially enriched vesicular proteins of interest were validated by Western blotting.
Atherosclerotic plaques were induced in wildtype mice through adenoviral PCSK9 overexpression and a 15-week high fat diet. After sacrifice, carotid arteries were extracted and mounted on to an ex-vivo perfusion system and subsequently perfused with fluorescently labelled EV to visualize their temporospatial distribution via multi-photon microscopy.
Results: Among 19 differentially expressed miRNAs, including nine upregulated and ten downregulated transcripts, miR-7-5p and miR-423-5p were found to be upregulated in EV derived from CXCL4-differentiated PBMCs compared to EV from M-CSF-differentiated PBMCs. The ongoing validation of these results by qRT-PCR confirmed the archival results with statistical and borderline statistical significance, respectively.
Besides seven other proteins, CD47 and CXCL4 were detected to be upregulated in EV derived from CXCL4-differentiated PBMCs.
In murine carotid arteries, migration of EV through myoendothelial projections, communication channels between vascular intima and media, was observed. Furthermore, EV were found to accumulate in atherosclerotic plaque areas.
Conclusion: Several miRNAs and proteins were found to be differentially enriched in CXCL4-differentiated PBMC-derived EV. The effects of their secretion on other cell types in the atherosclerotic plaque bear the potential to gain further mechanistic insight into pathogenic EV-based immune cell-matrix crosstalk in the context of vascular inflammation. The upregulation of CXCL4 strongly suggests a positive feedback loop as a driver of milieu-specific PBMC differentiation. The colocalization of EV with myoendothelial projections in murine atherosclerotic arteries indicates a potential role of these structures in the migration of circulatory EV into the vessel wall.
A Cross-section of murine carotid artery, EV (red) migrating into the vessel wall. Heat maps of differentially expressed proteins (
B) and miRNAs (
C)
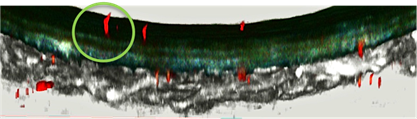 A
A
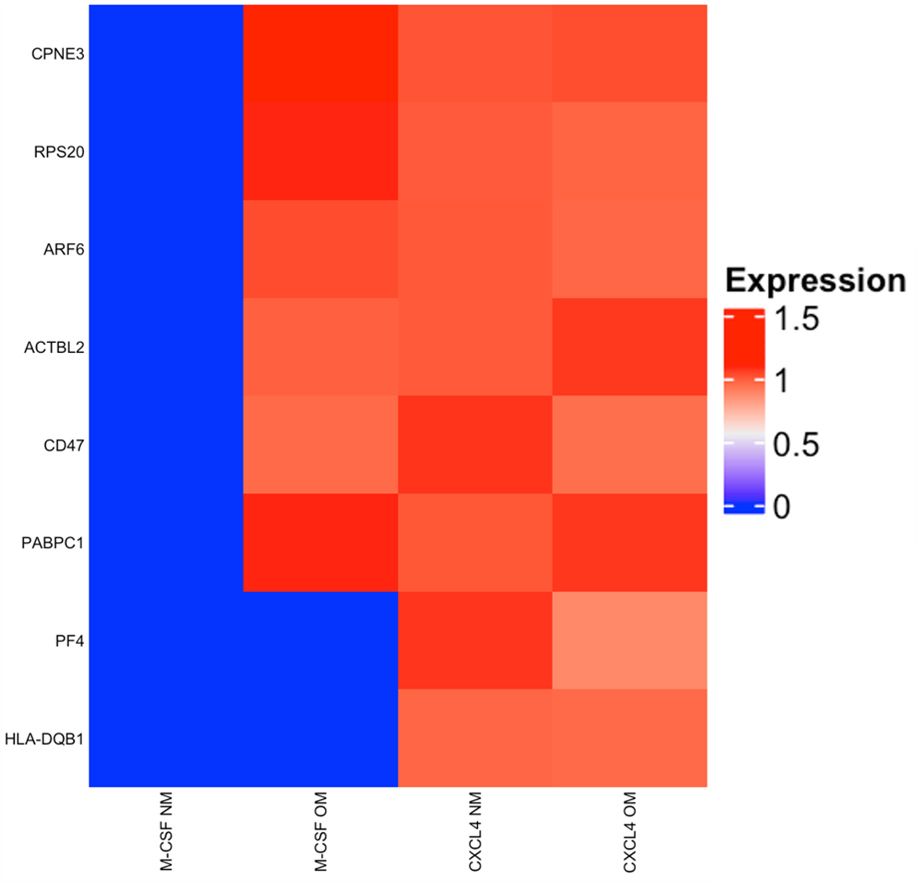 B
B 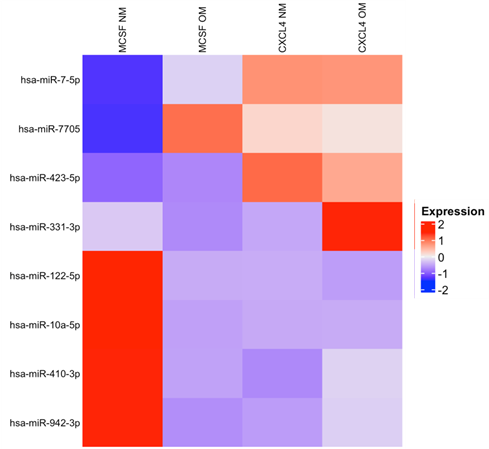 C
C
