Background: Infective Endocarditis (IE) is caused by microbial colonization of heart valve tissue associated with high morbidity and mortality rates. Current diagnostic algorithms rely on the modified Duke criteria including blood cultures and imaging modalities, e.g., transesophageal echocardiography (TEE). Biomarkers for early detection or verification of IE are not addressed in current guidelines.
Objectives and Methods: We aimed to identify diagnostic biomarkers for IE through neutrophil-centered immunophenotyping focusing on complement activation markers. We enrolled 64 patients with confirmed bacteremia and clinical suspicion of IE who underwent TEE as part of their diagnostic evaluation. Blood samples for flow cytometry (FC) were collected at the time of clinical suspicion. The primary goal was to discriminate patients with later confirmed IE from those with bacteremia but without endocarditic involvement. Our FC panel focused on classical neutrophil activation markers (e.g., CD11b, CD63, CD62L), complement receptor expression (e.g. C3aR, C5aR1), and reverse tissue migration (e.g. CD184, CD54). Furthermore, markers of systemic complement activation were measured in patient plasma.
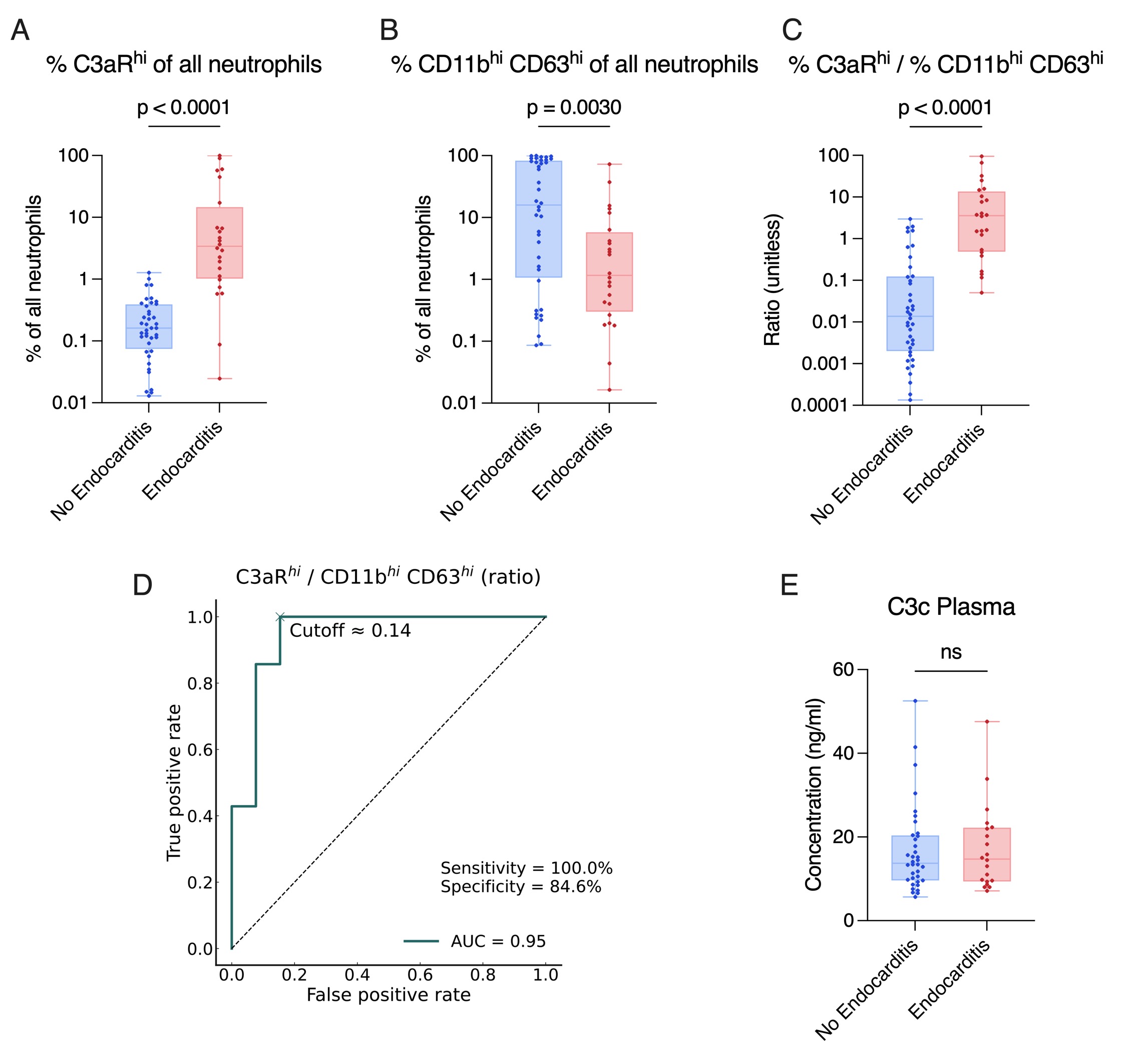
Results: IE was confirmed in 37.5% (n=24) of enrolled patients, while in 62.5% (n=40) of patients IE was excluded. As expected, relative and absolute neutrophil counts, along with CRP and PCT levels, showed no significant differences between the two groups.
Through deep immunophenotyping of neutrophils, we observed enhanced C3aR expression on neutrophils in IE patients (A), in contrast to neutrophil populations with lower levels of classical activation markers (CD11b, CD63) (B). We examined a significant difference between IE and non-IE patients through combining enhanced complement receptor expression with decreased classical activation receptor expression on neutrophils (C). Therefore, we created multiple population ratios, all indicative of IE presence, which allowed us to predict the condition with high sensitivity and specificity (D). Importantly, analysis of systemic complement activation, using a C3c-ELISA, showed no significant difference between IE and non-IE patients (E).
Conclusions: Our results suggest that continuous exposure of circulating neutrophils to vegetation material triggers localized complement receptor upregulation rather than a systemic activation. Accordingly, systemic complement activity, assessed by plasma C3c, did not differ between IE and non-IE patients, indicating a neutrophil-specific effect regarding upregulated complement receptor expression.
Taken together, complement receptor expression on neutrophils may serve as a novel, IE-specific biomarker, particularly useful in cases with inconclusive clinical or imaging findings. Our data provide first evidence supporting biomarker-assisted diagnostics of IE through complement receptor–based neutrophil phenotyping, potentially improving early detection and management.