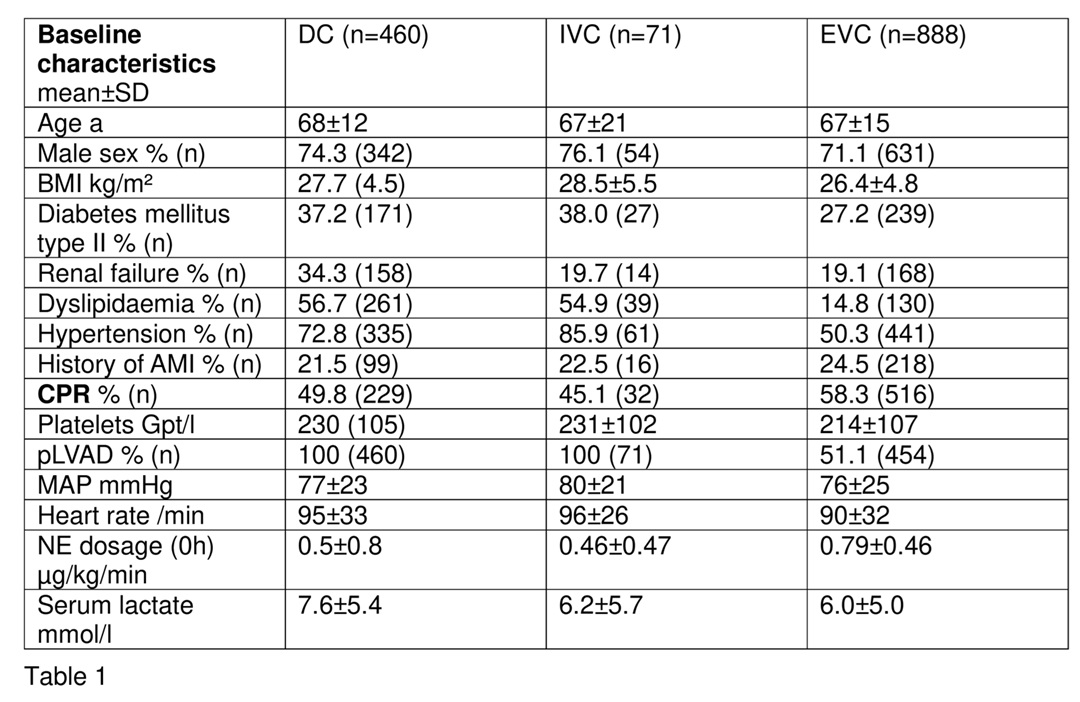
Background: The utilization of micro-axial percutaneous left ventricular assist devices (ma-pLVAD) showed an improved outcome in patients with cardiogenic shock (CS) complicating acute myocardial infarction. However, these devices are also used in patients with CS due to other causes und are associated with high material and personal costs. Therefore, a precise assessment of prognosis is of immanent interest to determine upfront, who might benefit from the device. Previously, we reported a novel 0 to 17 point risk score developed by using a hierarchical testing approach (Figure 1).
Purpose: We validated the novel scoring system predicting 30-day all-cause mortality specifically for CS patients treated with ma-pLVAD.
Methods: For internal validation data were collected from the ongoing Impella registry which was used for the development of the novel score. CS patients treated with ma-pLVAD (n=73), who were included in the registry after finishing score building, were used for internal validation (IVC). The cohort used for score building is called derivation cohort (DC) in the following (n=460). A large multicentric cohort of CS patients (n=888) was used for external validation (EVC). All-cause mortality was assessed by Kaplan-Meier method. Area under the curve (AUC) based on receiver operating characteristic (ROC) was estimated to determine the predictive value of the novel score in different cohorts. Continuous parameters are given as mean ± SD.
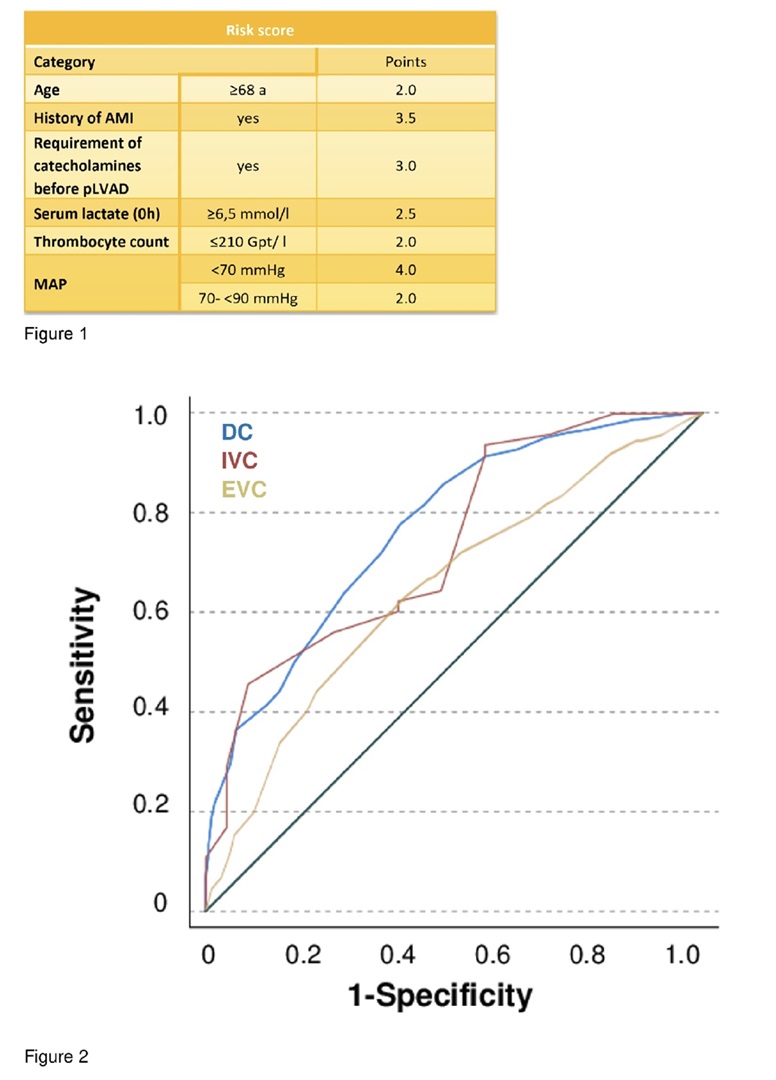
Results: Patients of the DC were 68±12 years old. In the IVC und the EVC the average age was 67±21 years and 67±15 years, respectively. Male gender was predominant in all cohorts (DC 74.3% [n=342], IVC 76.1% [n=54], EVC 71.1% [n=631]). A cardio-pulmonary resuscitation before ma-pLVAD was performed in 49.8% [n=229] in the DC, in 45.1% [n=32] in the IVC, and 58.3% [n=516] in the EVC. Initial serum lactate was 7.6±5.4 mmol/l in the DC, 6.2±5 .7 mmol/l in IVC, and 6.0±5.0 mmol/l in the EVC. All patients of the DC and the IVC received mechanical circulatory support with a ma-pLVAD, whereas only 51.1% (n=454) of the patients of EVC were treated with a pLVAD. Further baseline characteristics are shown in Table 1. All-cause mortality at 30 days was 65±2% in the DC, 68±5% in the IVC, and 55± 5% in the EVC. ROC analysis indicated good predictive capability of the novel score for 30-day-all-cause mortality in the DC (ROC AUC 0.767) and in the IVC (ROC AUC 0.736) as well. However, the score performance was lower in the external validation (ROC AUC 0.646; Figure 2).
Conclusion: We developed a clinically feasible scoring system to estimate 30-day-mortality of CS patients treated with ma-pLVAD with good internal validation. The lower predictive capability in the external validation might be driven by differences in baseline characteristics and treatment underlining the complexity of assessment of the prognosis in an all-comer CS cohort.