Objective:Valve-in-valve (ViV) transcatheter aortic valve implantation (TAVI) has emerged as a well-established treatment option for structural degeneration of surgical or transcatheter bioprosthetic aortic valves. Previous studies have highlighted several prognostic determinants in ViV patients, including left ventricular ejection fraction (LVEF), residual gradients, prosthesis–patient mismatch (PPM), renal dysfunction, and systemic comorbidities such as liver disease and pulmonary hypertension. However, the impact of residual aortic regurgitation (AR) post-ViV remains underexplored. This study aimed to identify independent predictors of mortality after ViV-TAVI in a contemporary long-term cohort.
Methods:This retrospective single-center cohort analysis included 231 adult patients who underwent ViV-TAVI between January 2010 and July 2025. Baseline echocardiographic and procedural data were collected, and follow-up was complete (median 24.8 months). Univariate and multivariate Cox regression as well as Kaplan–Meier survival analyses were performed.
Results:Median age was 80 years, 49.8% were female, and the median EuroSCORE II was 10.4. The median time from initial valve implantation to ViV was 9 years [IQR 6–12]. Common comorbidities included hypertension (91.3%) and coronary artery disease (56.3%). Post-ViV aortic regurgitation, including valvular and paravalvular components, was observed in 69 patients (30%), predominantly grade 1 (n = 63). This regurgitation was identified as a significant independent predictor of late mortality (HR = 2.12, p = 0.016), with survival curves diverging after 12 months. In contrast to earlier reports emphasizing post-procedural gradients and PPM as key prognostic factors, neither pre- nor post-ViV mean gradient was associated with mortality when analyzed categorically or continuously. LVEF remained a strong independent predictor (HR = 0.95 per 1% increase, p < 0.001), confirming prior findings in TAVI populations. EuroSCORE II correlated significantly with long-term mortality (p < 0.001), with each 1% increase predicting a 1.98% rise in mortality risk. The type of initial valve replacement (SAVR vs TAVI) showed no independent influence on survival (SAVR n = 198, TAVI n = 33; p = 1.0000).
Conclusion:In this ViV-TAVI cohort, residual aortic regurgitation—although routinely assessed after the procedure—emerged as a previously underrecognized yet decisive predictor of late mortality. Unlike residual gradients, which showed no prognostic relevance, AR demonstrated a clear and independent association with adverse long-term outcomes. These findings underscore the need to interpret even mild post-ViV AR with greater clinical attention. The type of initial valve replacement—surgical or transcatheter—showed no significant effect on survival. Alongside other parameters, AR should be regarded as a key factor for postprocedural risk stratification and follow-up optimization in ViV-TAVI patients.
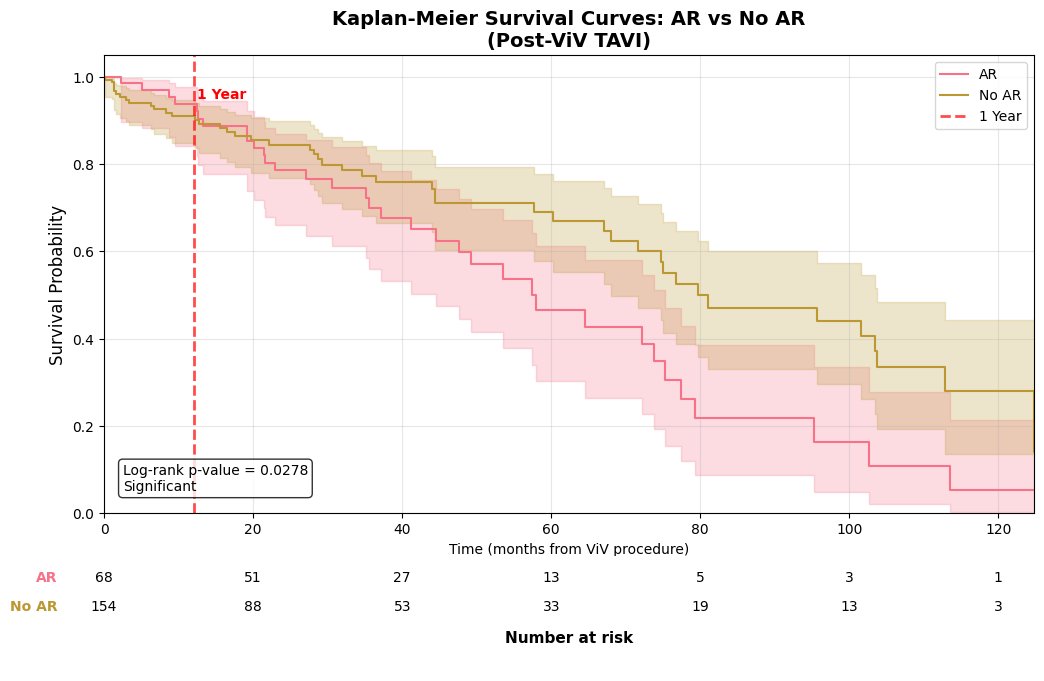
Kaplan–Meier survival after ViV‑TAVI stratified by post‑procedural aortic regurgitation (AR vs no AR). Survival was lower in the AR group (n=68) than in the no‑AR group (n=154); log‑rank p=0.028. The table lists numbers at risk at 20‑month intervals aligned with the x‑axis.
