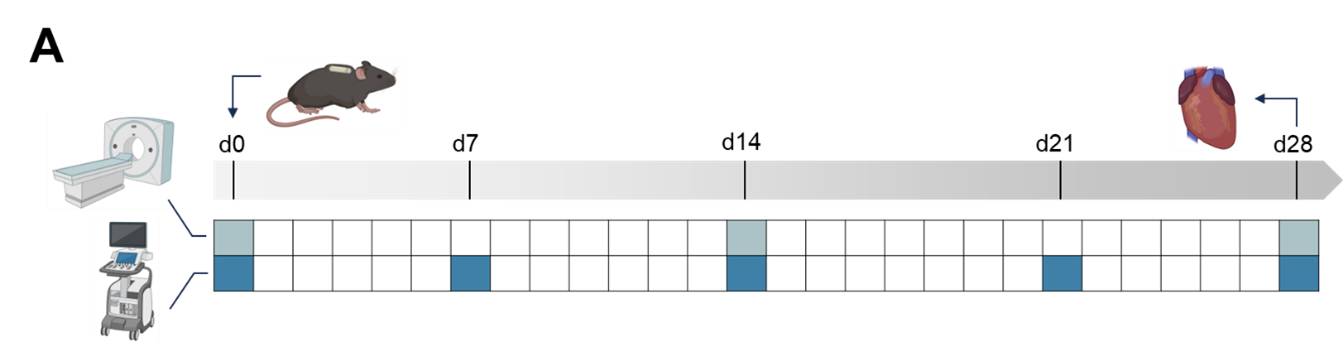Background and AimsMetabolic syndrome and heart failure (HF) represent major global health challenges, driven in part by impaired cardiac energy metabolism. Sodium-glucose cotransporter 2 (SGLT2) inhibitors, originally developed for type 2 diabetes, have demonstrated profound cardiovascular benefits independent of glycemic control (DAPA-HF, EMPEROR-REDUCED, EMPEROR-PRESERVED, DELIVER studies). Their mechanisms are thought to involve restoration of metabolic homeostasis through promoting mitochondrial function and shifting cellular signaling from nutrient surplus (via mTOR) to nutrient deprivation pathways (via AMPK). Cyclophilin A (CyPA), a ubiquitously expressed chaperone that can be secreted under cellular stress, has been implicated in cardiac remodeling and HF progression through its interaction with its receptor EMMPRIN (CD147). Evidence suggests that CyPA may regulate metabolic signaling pathways, including AMPK and glucose utilization. We hypothesized that inhibition of extracellular CyPA may beneficially modulate cardiac metabolism in HF, potentially mimicking some metabolic effects observed with SGLT2 inhibition.
MethodsAtherosclerosis-prone ApoE
-/- mice were treated with Angiotensin II (Ang II) to induce chronic HF and received either a monoclonal antibody against extracellular CyPA (α-CyPA) or IgG control via osmotic minipumps. Control groups received NaCl. Cardiac function and metabolism were assessed longitudinally using weekly echocardiography and [
18F]FDG-PET-MRI at three time points. Heart and plasma samples were collected for metabolomic (NMR) and histological analyses. Metabolic alterations in the diseased myocardium were studied spatially resolved via mass spectrometry imaging (MSI). Planned follow-up studies include cardiomyocyte glucose uptake assays, mitochondrial function analysis, and targeted protein signaling experiments.
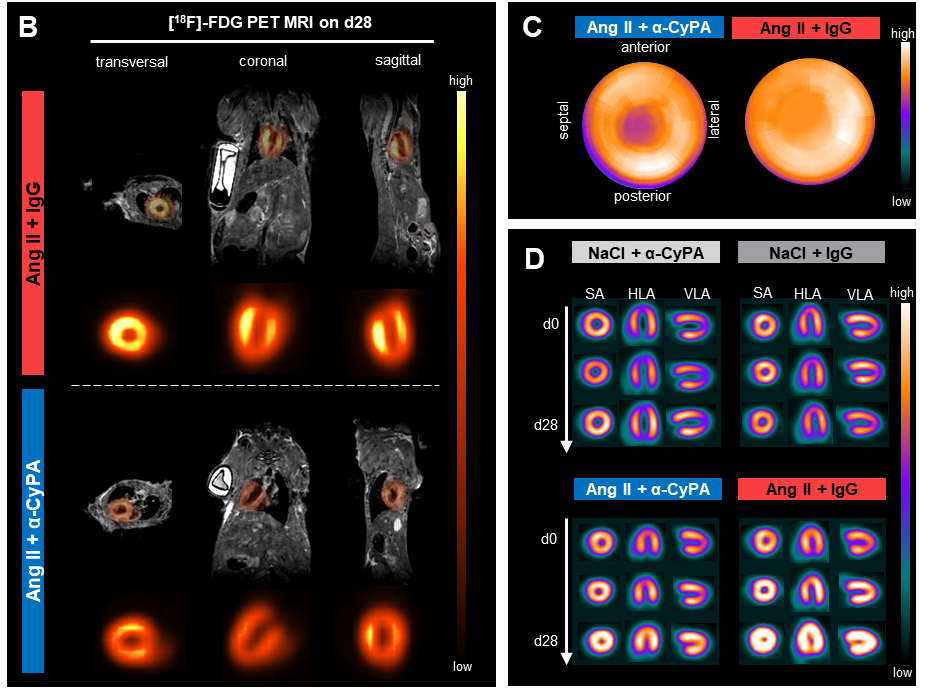
ResultsInhibition of extracellular CyPA markedly reduced glucose uptake in the myocardium of Ang II-treated mice, an effect not previously described in this HF model. Ang II + IgG animals displayed both increased cardiac FDG uptake and reduced blood glucose levels, suggesting enhanced pathological myocardial glucose sequestration. Preliminary analysis revealed a correlation between increased cardiac glucose uptake and impaired left ventricular systolic function. Notably, inflammation was low in this model, indicating that CyPA may directly influence metabolic regulation rather than acting solely through inflammatory pathways, which has been proposed before.
ConclusionOur findings suggest that extracellular CyPA contributes to maladaptive metabolic remodeling in HF by promoting excessive myocardial glucose uptake. Antibody-mediated CyPA inhibition may restore metabolic balance and preserve cardiac function. Ongoing mechanistic and metabolomic studies aim to elucidate the signaling pathways involved. Targeting extracellular CyPA may represent a novel therapeutic strategy to modulate cardiac metabolism in HF, providing potentially complementary clinical effects to those of SGLT2 inhibitors.