Background
Post-myocarditis ventricular tachycardia (VT) is a unique non-ischemic substrate marked by intricate scar architecture. Information on scar distribution, myocardial layer involvement, and ablation success rates is still limited.
Methods
We retrospectively examined 31 consecutive patients who underwent RF ablation for scar–related VT following myocarditis. Contrast-enhanced CT scans taken before the procedure were processed with the inHEART medical platform for 3D segmentation and scar analysis. Scar regions were identified by late enhancement and classified by location (septal, basal, mid-ventricular, apical, anterior, inferior, lateral) and myocardial layer (endocardial, midmyocardial, epicardial). Wall thinning was evaluated separately. The main endpoint was recurrence of VT or ventricular arrhythmia (VA). An exploratory analysis compared VT/VA recurrence-free survival between this group and a cohort of patients with ischemic cardiomyopathy (ICM) who also underwent RF ablation with CT-guided image integration.
Results
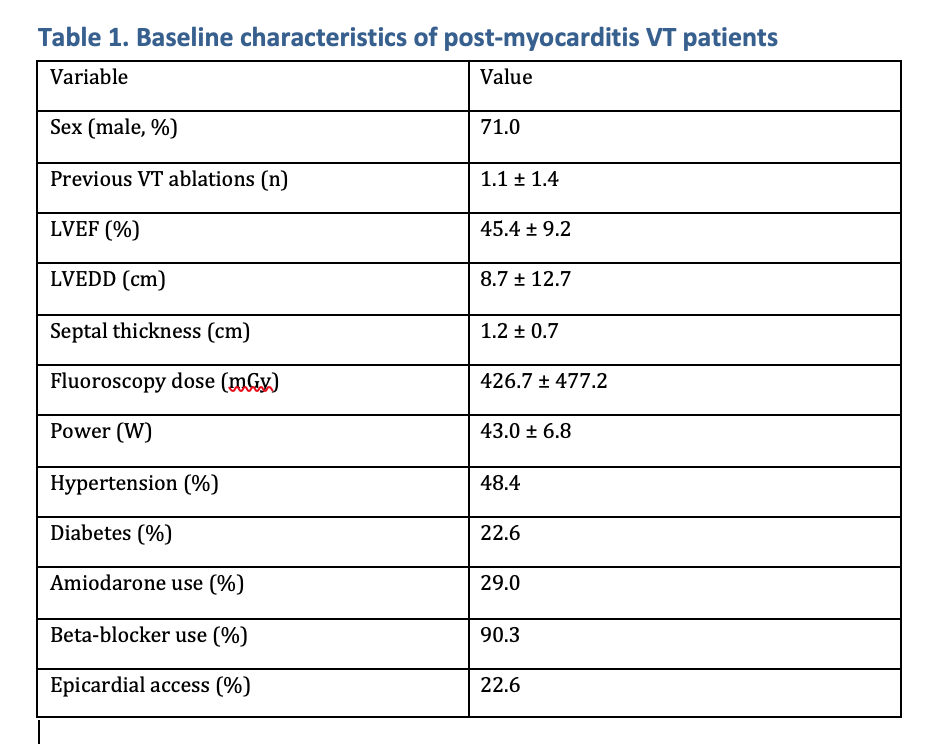
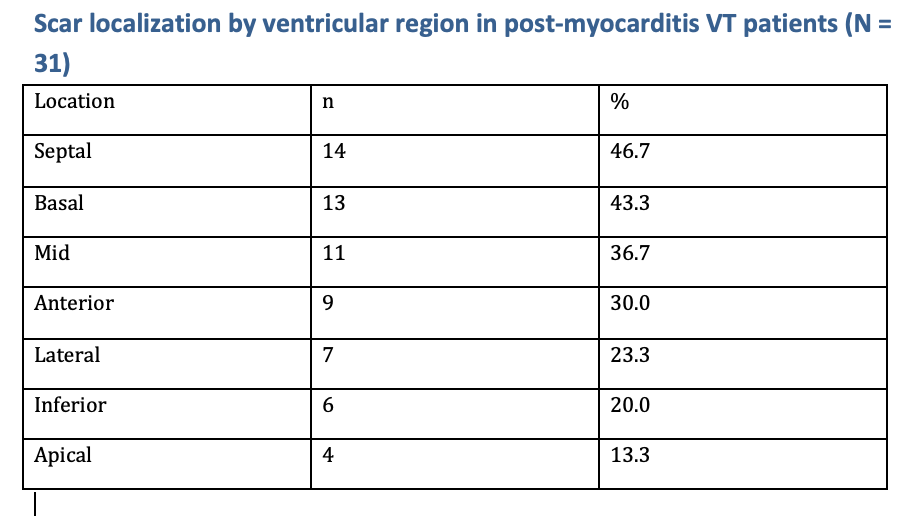
Most patients were male (71%) with moderately reduced LV function (LVEF 45 ± 9%) and LV dilatation (LVEDD 8.7 ± 0.8 cm). Hypertension and diabetes were present in 48% and 23%, respectively. Most (90%) of patients received β-blockers, and 29% amiodarone (Figure 1). Scar architecture was typically non-ischemic, with midmyocardial involvement in 87%, epicardial in 52%, and purely endocardial scar in 23%. Scars most often affected the septal (46%), basal (43%), and mid-ventricular (36%) regions. (Figure 2)
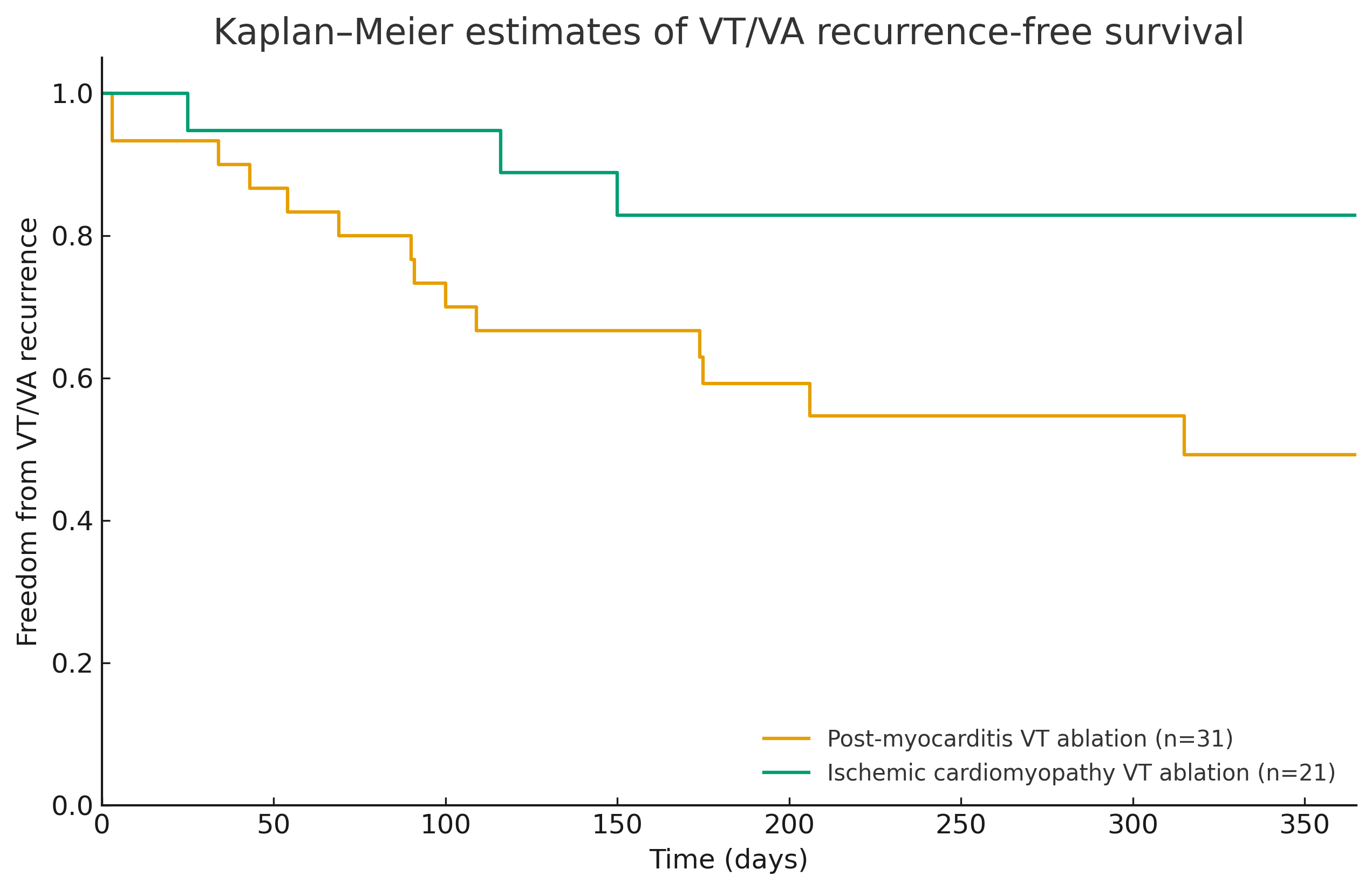
Over an average follow-up of 9.1 ± 7.1 months, VT/VA recurrence occurred in 15 out of 31 (48%) patients. Recurrence was mainly seen in patients with midmyocardial involvement (50%) and multilayer substrates. The pattern involving endocardial, midmyocardial, and epicardial layers had the highest recurrence rate (75%). The mid-ventricular midmyocardial scar was identified as the most problematic regional substrate. Compared to the ischemic cohort, post-myocarditis patients showed significantly lower VT/VA recurrence-free survival at 12 months (Figure 3) (p=0.04).
Conclusions
In post-myocarditis VT, inHEART-based CT segmentation reveals predominantly midmyocardial and epicardial, often multilayered, scar tissue clustered in the septal, basal, and mid-ventricular regions, associated with a high VT/VA recurrence rate within the first year after RF ablation. Increasing transmural and mid-ventricular substrate complexity identifies a high-risk phenotype, supporting the use of advanced image-guided substrate characterization and tailored ablation strategies, including consideration of intramural and epicardial targets, in this challenging population.