Background. Aortic stenosis (AS) is the most common valvular heart disease with rising prevalence. Despite successful valve replacement, myocardial fibrosis (MF) contributes significantly to adverse outcomes. While histological analysis remains the gold standard, it is unsuitable for routine use. Cardiac magnetic resonance imaging (CMR) offers non-invasive surrogates but has limited sensitivity for diffuse fibrosis and high resource requirements, which restrict its universal applicability. Therefore, we developed machine learning (ML) models to predict histology-proven MF using routinely available data as a first-line screening tool and evaluated their prognostic value.
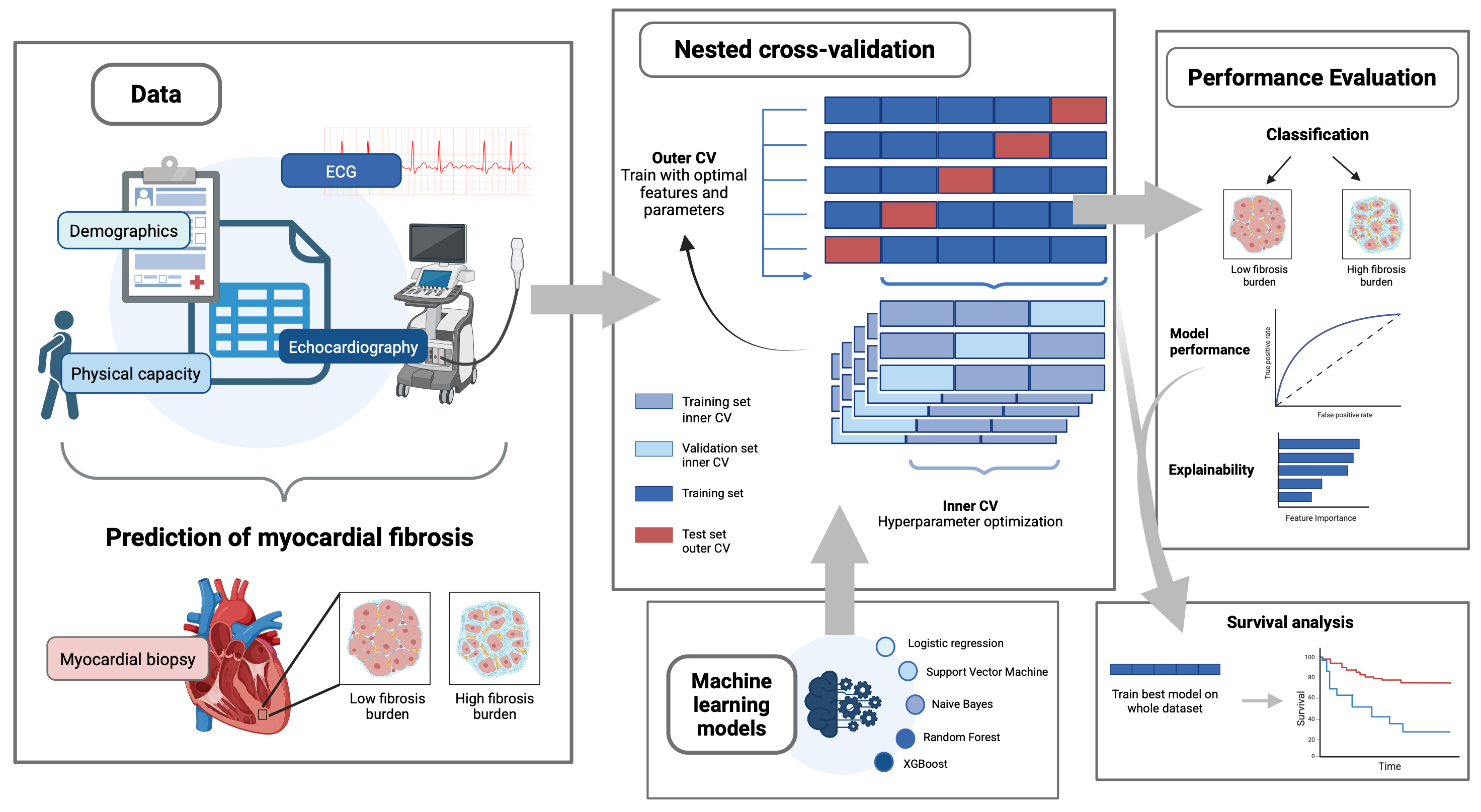
Methods. We prospectively enrolled 325 consecutive patients with severe AS undergoing TAVR (2017-2022). Of these, 155 patients underwent endomyocardial biopsy with histological quantification of fibrosis (≥11% collagen area). This cohort served for ML model development using nested stratified cross-validation (5 outer/3 inner folds). We evaluated five ML algorithms (logistic regression, support vector machines (SVM), random forest, XGBoost, and naive Bayes) using 24 prespecified predictors across clinical, ECG, and echocardiographic domains, including regional longitudinal strain from segments adjacent to the biopsy site. The remaining 170 patients formed an external validation cohort for prognostic assessment via Kaplan-Meier survival analysis. The study workflow is demonstrated in Figure 1.
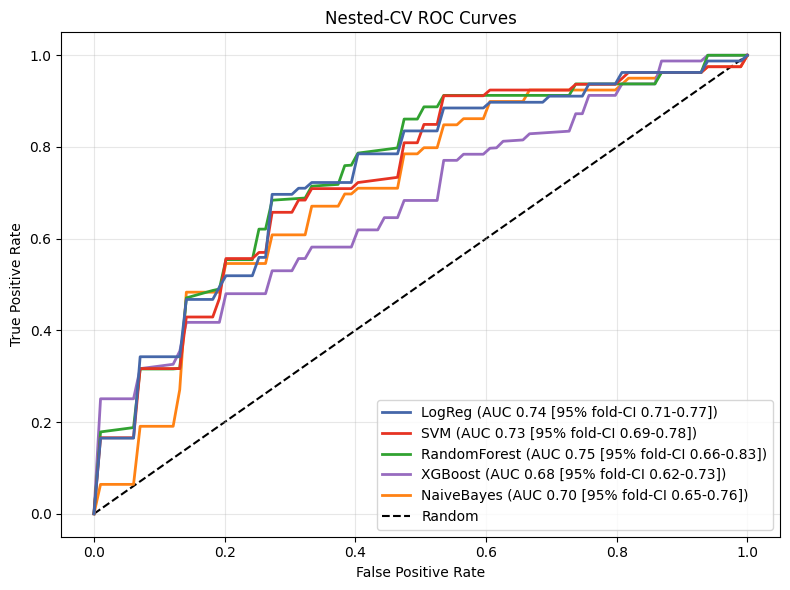
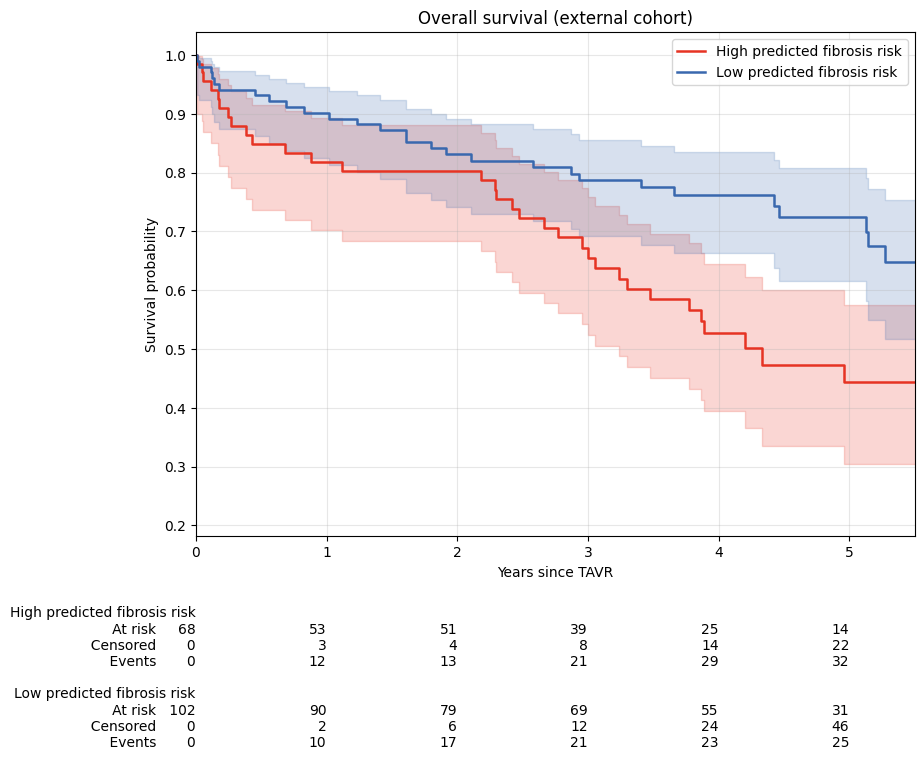
Results. Mean age was 79.5±6.3 years, 43% of the patients were female, and 66% of them were in NYHA class III/IV. In the biopsy cohort, 79 patients (51%) had high fibrotic burden. Compared to patients with low fibrosis, those with high fibrosis showed reduced functional capacity (6-minute walk: 171 vs. 245m, p=0.019), larger LVEDV (96±47 vs. 78±32ml, p=0.008), lower LVEF (47±15% vs. 54±11%, p<0.001), and impaired GLS (-14±5 vs. -16±4, p=0.002). Across machine learning models, the random forest model achieved the best discrimination (AUC 0.75, 95%CI 0.66-0.83) with a high sensitivity of 0.80 and moderate specificity of 0.70 (Figure 2). The subsequent SHAP analysis identified LVEDD, regional strain segments, 6-minute walk distance, BMI, and TAPSE as top predictors. In the external validation cohort, ML-predicted high-risk patients exhibited significantly worse overall survival, with a 5-year overall survival rate of 44% compared to 72% (p=0.004); see Figure 3.
Conclusions. ML models using routine clinical and echocardiographic data predict histology-proven myocardial fibrosis with good discrimination and demonstrate strong prognostic value for long-term mortality after TAVR. This non-invasive risk stratification could serve as a first-line screening tool to identify high-risk patients who may benefit from advanced imaging and targeted antifibrotic therapies.