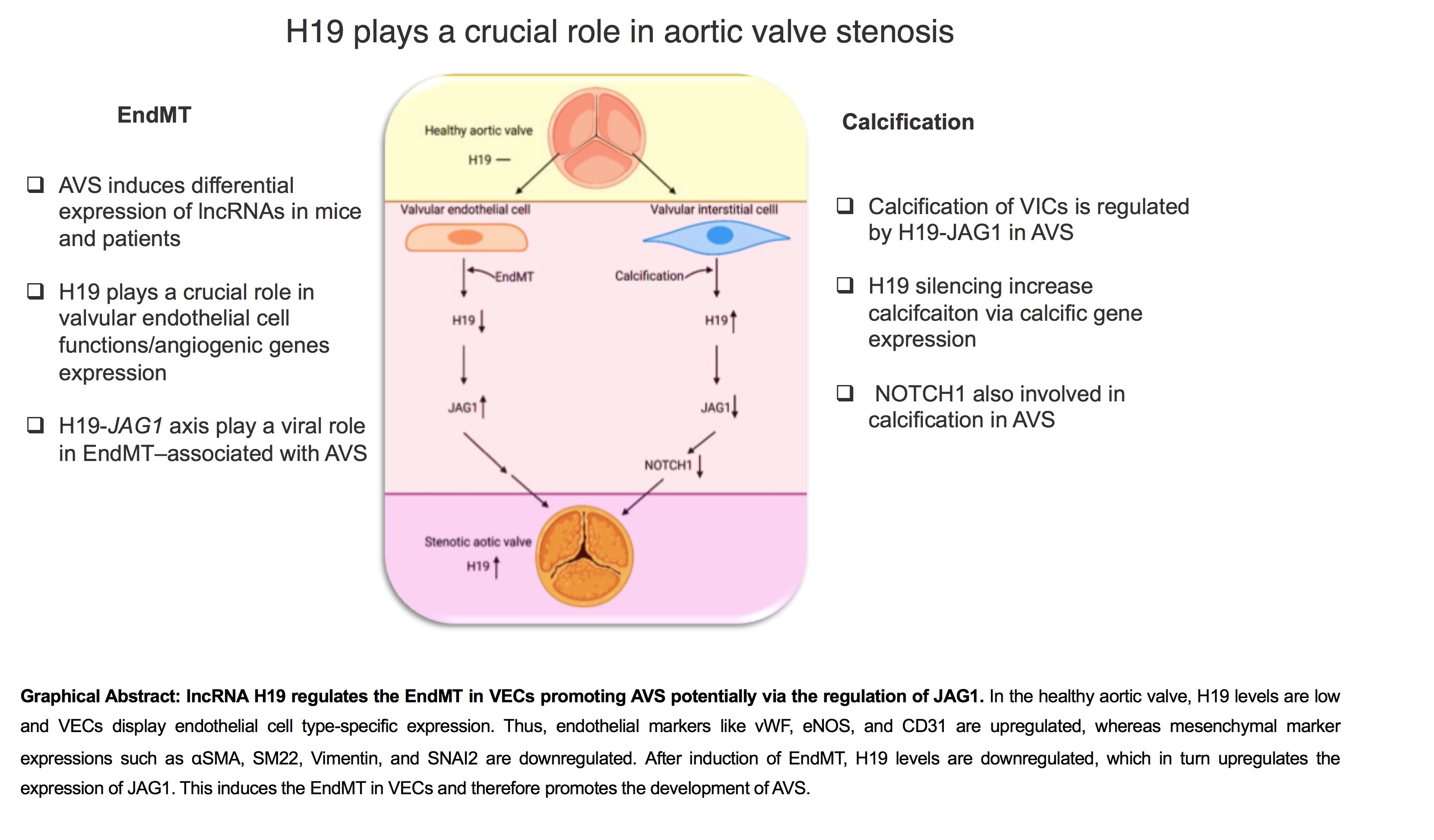
Background: Aortic valve stenosis (AVS) is a life-threatening condition with no pharmacological interventions. Endothelial-to-mesenchymal transition (EndMT) represents a fundamental mechanism driving disease progression, where valvular endothelial cells (VECs) undergo pathological transformation. While long noncoding RNAs (lncRNAs) regulate cardiovascular pathogenesis, their specific role in EndMT during AVS remains undefined. We identified the lncRNA H19 as a significantly dysregulated molecule in human stenotic valves and investigated its functional role in EndMT-mediated AVS pathogenesis.
Methods and Results: RNA sequencing of human aortic valve tissues from AVS patients revealed profound H19 upregulation compared to non-diseased controls. Using primary human VECs under EndMT-promoting conditions, we demonstrated that H19 knockdown potently accelerated the EndMT process, as evidenced by suppressed endothelial markers (vWF, NOS3) and induced mesenchymal markers (αSMA, SM22, Vimentin, SNAI2) at both transcriptional and protein levels. Functional characterization revealed that H19 silencing enhanced VEC migration and disrupted angiogenic sprouting capacity. Mechanistic investigation through an angiogenesis PCR array identified JAG1 as the most significantly dysregulated target (4.43-fold downregulation upon H19 loss). Subsequent validation confirmed H19-dependent JAG1 regulation and implicated NOTCH1 signaling and STAT3 activation in this pathway. Importantly, pharmacological STAT3 inhibition reversed the pro-EndMT effects of H19 knockdown, establishing a definitive H19-JAG1-STAT3 signaling axis driving EndMT in AVS.
Conclusion: Our findings establish lncRNA H19 as a master epigenetic regulator of EndMT in aortic valve stenosis. We delineate a novel pathogenic pathway where H19 drives VEC transformation through coordinated regulation of JAG1/NOTCH1 signaling and STAT3 activation. This work not only reveals fundamental insights into AVS pathogenesis but also identifies H19 and its downstream effectors as promising therapeutic targets for a disease with no current medical treatment options.