Background:
Patients with degenerative high-grade aortic stenosis (AS) who develop symptoms require timely valve replacement to prevent rapid progression to heart failure and death. Structural myocardial changes such as left ventricular hypertrophy and diastolic dysfunction may also occur in transthyretin cardiac amyloidosis (ATTR-CA), a disease prevalent in elderly individuals and associated with adverse outcomes. Computed tomography (CT) is routinely performed prior to transcatheter aortic valve replacement (TAVR). A standard photon-counting CT (PCCT) protocol can be readily adapted to include quantification of myocardial extracellular volume (ECV). While moderate ECV elevation is common in severe AS, markedly increased ECV values strongly suggest coexisting ATTR-CA, particularly when light-chain amyloidosis has been excluded by serum and urine testing. An ECV threshold above 37% has been proposed to identify cardiac amyloidosis with high sensitivity and specificity. We aimed to assess the prevalence of potential coexistent ATTR-CA in elderly AS patients undergoing TAVR using a modified CT protocol incorporating myocardial ECV analysis.
Methods and Results:
A total of 174 consecutive patients (mean age 81 ± 6 years, 59% men) with symptomatic severe AS scheduled for TAVR were prospectively enrolled. All underwent photon-counting CT (Naeotom Alpha, Siemens Healthineers) including an additional delayed cardiac acquisition 5 minutes after the standard aortic run-off TAVR-CT. Pre-contrast and delayed datasets were used to calculate myocardial ECV.
Mean ECV was 29 ± 6%. In 8 patients (5%), ECV exceeded 37%, indicating possible coexistent cardiac amyloidosis.
Conclusions:
Integration of a brief delayed cardiac acquisition into a standard TAVR CT protocol allows quantification of myocardial ECV and may serve as a convenient non-invasive method to screen for concomitant cardiac amyloidosis in patients with severe AS. As coexistent amyloidosis negatively influences outcomes after TAVR, further diagnostic work-up including exclusion of light-chain disease and 99mTc-DPD scintigraphy is warranted in patients with ECV > 37%.
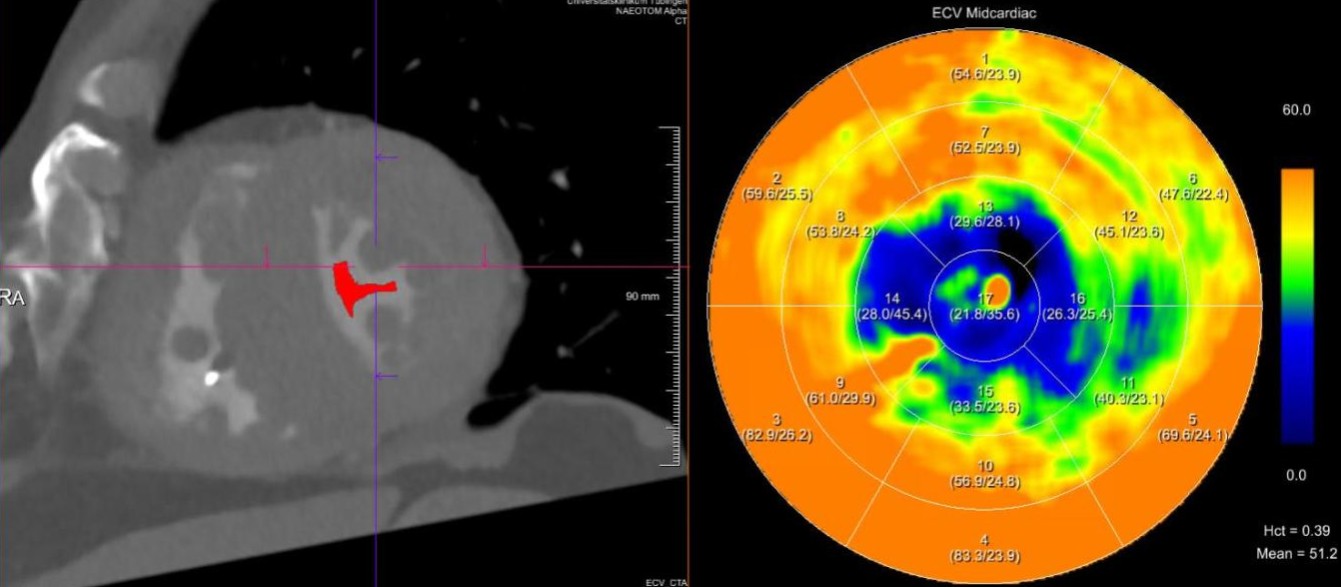
Figure legend:
An 85-year-old male with symptomatic high-grade AS referred for TAVR. A modified CT protocol with delayed cardiac acquisition revealed a markedly increased ECV of 58.9%. The CT short-axis image (left) and ECV-derived AHA 17-segment polar map (right) demonstrate diffusely elevated ECV sparing only the apex, consistent with cardiac amyloidosis.