BackgroundThe development and sustainability of atrial arrhythmias, like atrial fibrillation (AFib), rely on proarrhythmic cardiac remodeling, encompassing functional, electrical and structural changes. Structural atrial remodeling involves formation of fibrotic patches as well as atrial dilatation, thereby establishing a proarrhythmic substrate. This process facilitates heterogenous propagation of atrial signals and enables reentry circuits, which are hallmarks of AFib. Metabolic diseases, like diabetes mellitus, are key risk factors for AFib, but little is known about the mechanisms that drive the formation of an atrial proarrhythmic substrate.
PurposeTo investigate underlying mechanisms leading to proarrhythmic atrial remodeling in diabetes mellitus using a transgenic swine model (INSC94Y).
MethodsSix-month-old swine of either sex (16 wild-type (WT) and 22 diabetic INSC94Y (DM)) were sedated, intubated and mechanically ventilated. Atrial diameter was determined using echocardiography. Subsequently, invasive electrophysiology studies were performed and the susceptibility to atrial arrhythmias was determined by burst pacing. The heart was excised and atrial tissue samples were collected. Atrial fibrosis was assessed using a Masson-Trichrome staining. Snap-frozen atrial free-wall and appendage samples were analyzed on a proteome level using LC-MS/MS (N=5). Differentially expressed genes (P<0.05) were submitted to the STRING database.
ResultsDM swine showed increased blood glucose levels (107 mg/dL (WT) vs 347 mg/dL (DM); p<0.0001). The inducibility of AFib in this group was significantly higher compared to WT (figure 1-A, B). Echocardiographically, no differences in atrial dimensions were observed (figure 1-C). Histological analysis indicated significantly more atrial fibrosis in DM swine both in the left atrial free-wall (figure 1-D) and left atrial appendage (figure 1-E). In order to further understand the mechanisms behind atrial fibrosis formation, the atrial proteome was analyzed. STRING network analysis indicated that the top-10 overrepresented processes were strongly associated to extracellular matrix maintenance and mitochondrial function (figure 1-F, G, H).
ConclusionsThis study aimed to elucidate structural remodeling processes leading to AFib using a transgenic diabetic swine model. As a marker of structural remodeling we observed significant atrial fibrosis. However, gross anatomical changes, like atrial dilatation, were absent. As differentially expressed proteins in DM atria were strongly associated to ECM modification and mitochondrial function, future studies on the role of these processes in atrial remodeling are warranted to identify potential targets for novel therapeutic approaches.
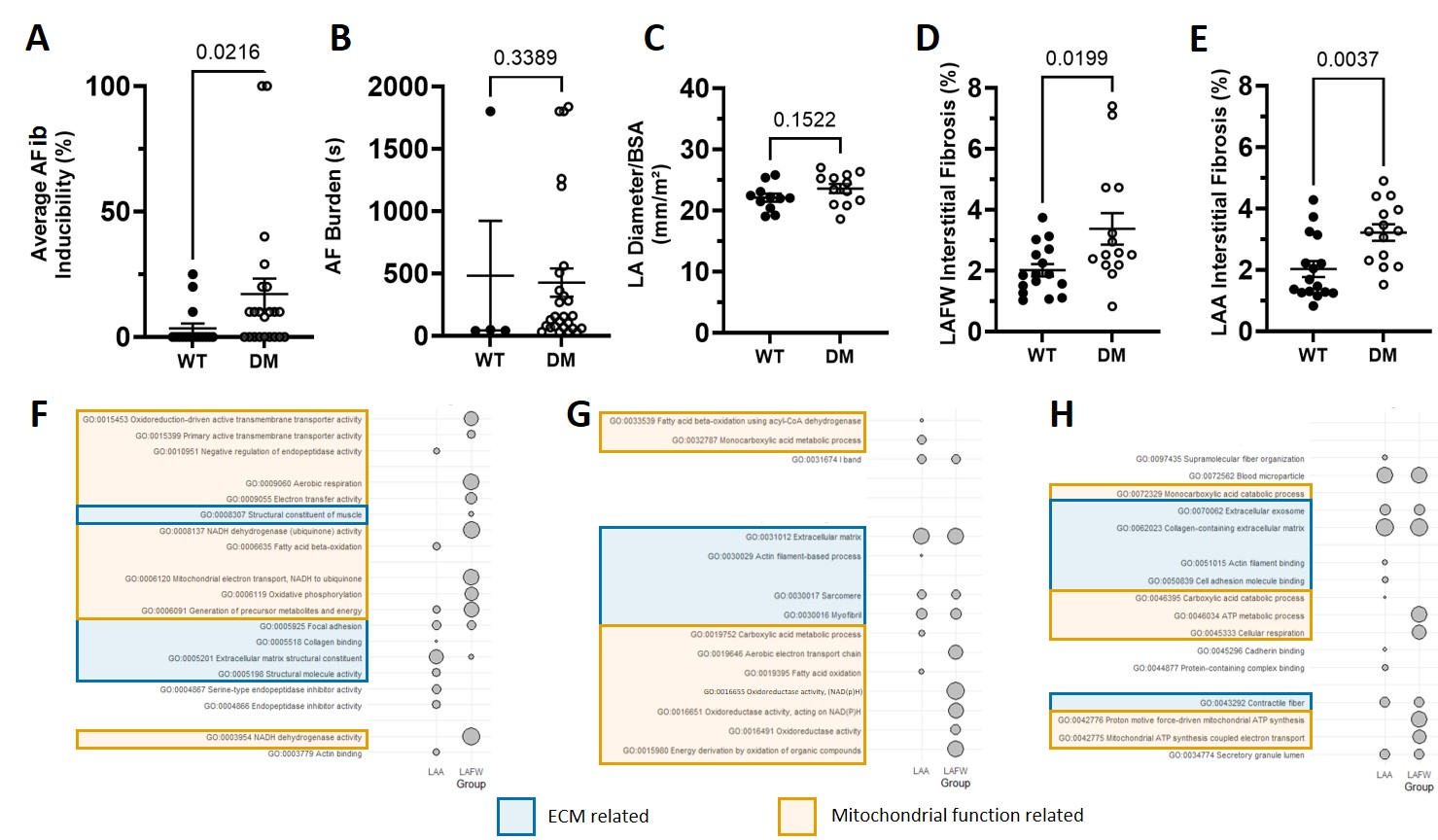
Figure 1: (A) Inducibility of AFib in wild-type (WT) and diabetic (DM) swine. (B) Cumulative AFib burden (s) for each swine. (C) LA dimension, corrected for body surface area (BSA) (mm/m2). (D) Quantification of fibrosis in the left-atrial free wall (LAFW) using a Masson-Trichrome staining. (E) Quantification of fibrosis in the left-atrial appendage (LAA) using a Masson-Trichrome staining. (F, G, H) Strongest biological (F), cellular (G) and molecular (H) gene-ontology terms for LAA and LAFW, based on signal.
