Background: The cardiovascular–kidney–metabolic (CKM) framework integrates systemic metabolic and renal dysfunction as key drivers of cardiovascular risk, yet its prognostic utility following transcatheter aortic valve implantation (TAVI) remains underexplored. This study evaluated whether CKM phenotypes independently predict 36-month outcomes including all-cause mortality, cardiovascular (CV) death, and heart failure (HF) hospitalization in a real-world TAVI cohort.
Methods: A total of 1,275 patients (mean age 81±6 years; 52% male), who underwent TAVI between 2009 and 2017 in our center were enrolled in this study. Patients were divided in to following CKM phenotype group: no burden (n=352), metabolic-only (n=182), kidney-only (n=488), and combined burden (n=253). Metabolic burden included ≥2 of obesity, diabetes, or atherogenic dyslipidemia; kidney burden was defined as eGFR <60 mL/min/1.73m² or dialysis. The primary endpoint was all-cause mortality; secondary endpoints included CV mortality and HF hospitalization. Cox regression models were adjusted for age, sex, NYHA class III–IV, atrial fibrillation (AF), hypertension, hyperuricemia, coronary artery disease (CAD), and chronic obstructive pulmonary disease (COPD).
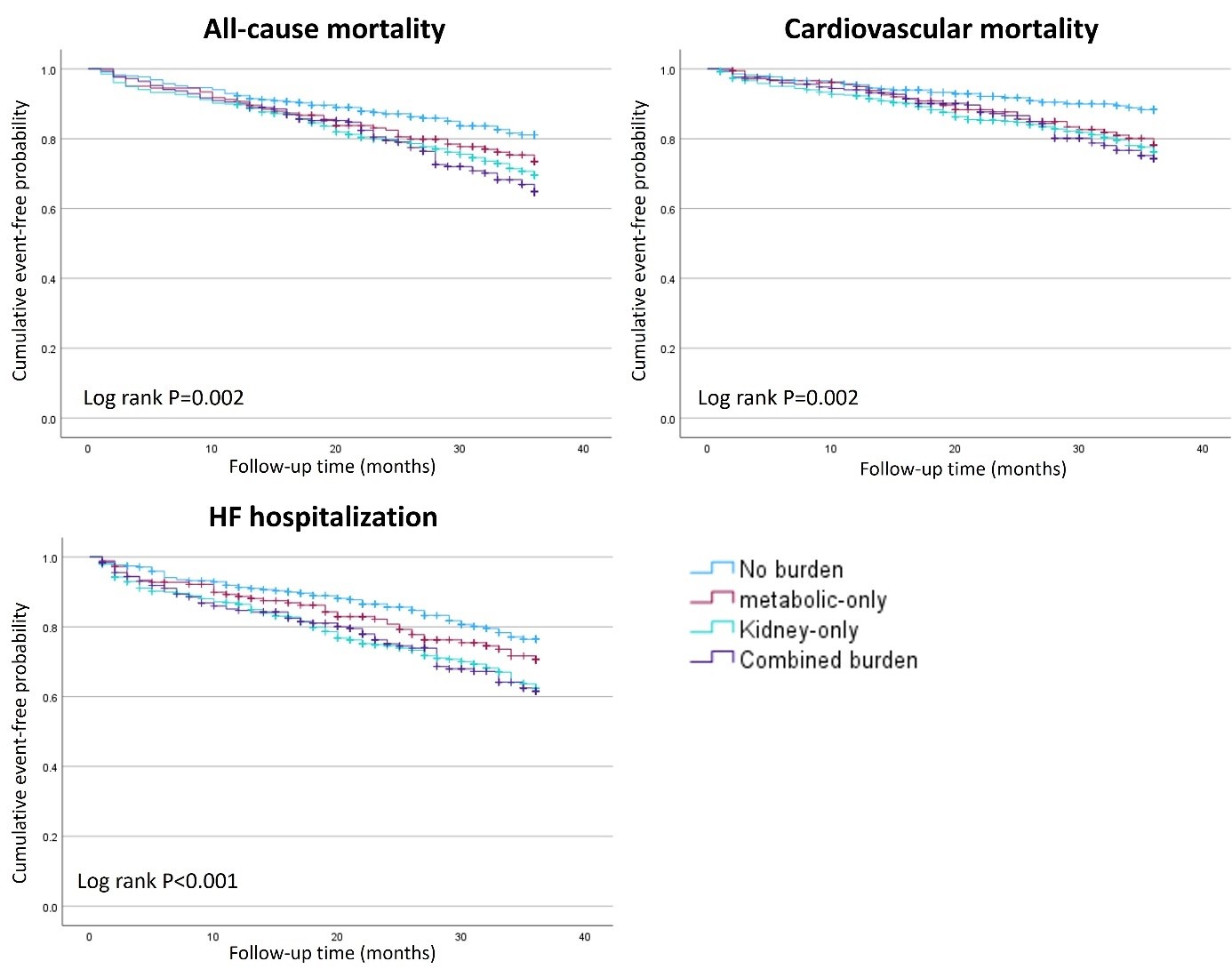
Results: During 36-month follow-up, 298 patients (23.4%) died, 209 (16.4%) from CV causes, and 325 (25.5%) were hospitalized for HF. Outcome rates increased stepwise with CKM burden. All-cause mortality ranged from 15.9% in the no-burden group to 29.2% in patients with combined burden. CV mortality was 9.7%, 18.1%, 18.9%, and 19.8% in no-burden, metabolic-only, kidney-only, and combined groups, respectively. Similarly, HF hospitalization ranged from 17.3% to nearly 30% across the gradient (log-rank p<0.001 for all outcomes). Multivariable analysis showed that CKM phenotypes remained as independent predictors for the primary and secondary outcomes. Compared to the no-burden group, hazard ratios for all-cause mortality were 1.68 (95% CI: 1.22–2.30, p=0.001) for kidney-only and 1.83 (1.29–2.60, p<0.001) for combined burden. CV mortality was elevated in all three CKM subgroups: 1.72 (1.06–2.79, p=0.028) for metabolic-only, 1.86 (1.25–2.77, p=0.002) for kidney-only, and 1.90 (1.22–2.95, p=0.005) for combined. For HF hospitalization, hazard ratios were 1.68 (1.24–2.27, p<0.001) and 1.65 (1.18–2.32, p=0.004) in kidney-only and combined groups, respectively. Atrial fibrillation consistently emerged as the strongest non-CKM covariate across all models (HR 1.70–1.91, p<0.001).
Conclusions: CKM phenotypes provide powerful, independent prognostic value for long-term mortality and HF risk following TAVI. Kidney dysfunction alone significantly increases adverse event rates, and its combination with metabolic abnormalities further amplifies this risk. Even metabolic burden alone conferred excess CV mortality. These findings validate the CKM framework as a practical and integrative approach for risk stratification post TAVI. Incorporating CKM phenotyping into pre- and post-TAVI evaluation may add patient selection, resource allocation, and longitudinal care planning.