Background
Pulmonary function testing (PFT) can elucidate cardiopulmonary interactions in acute heart failure (AHF), but its diagnostic and prognostic value remains uncertain. This study evaluates the diagnostic and prognostic utility of spirometry and diffusing capacity for carbon monoxide (DLCO) testing in acute decompensated patients and examines their associations with laboratory and echocardiographic parameters.
Methods
Consecutive patients hospitalized with AHF were prospectively offered spirometry and DLCO measurements using the portable EasyOne Pro device. Forced expiratory volume in one second (FEV₁), forced vital capacity (FVC), and DLCO (each in percent predicted) were quantified on admission and at discharge following guidelines from the European Respiratory Society (ERS). Associations between pulmonary and cardiac parameters (LVEF, TAPSE, E/E’, NT-proBNP, and NYHA class) were assessed using linear regression models. For survival analyzes, Firth-penalized Cox regression was conducted.
Results
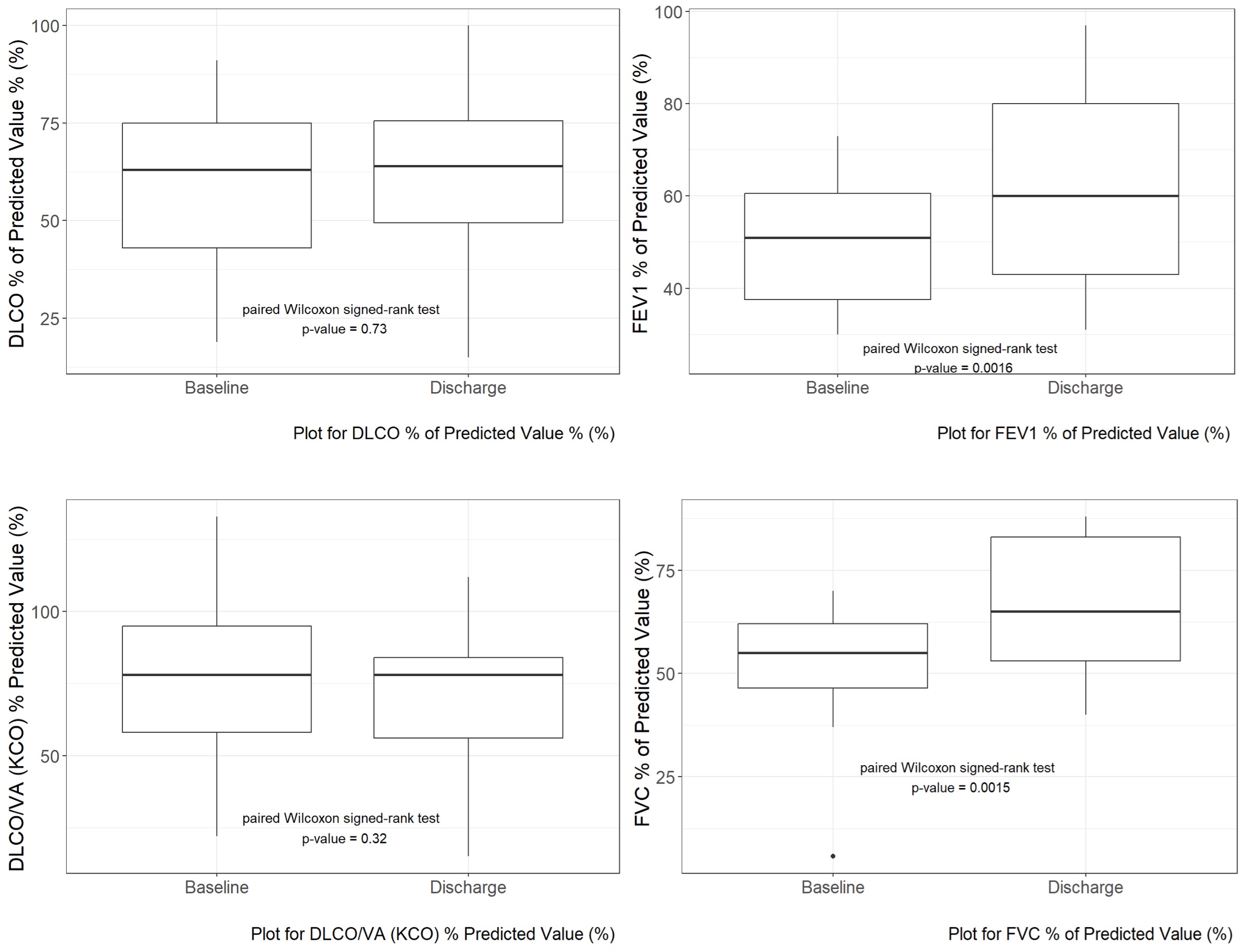
Sixty-one patients (median age was 70 years [IQR: 61-81]; 21.3% women) with a median left ventricular ejection fraction (LVEF) at baseline of 30% [IQR: 20-50] were enrolled. As most likely leading cause of cardiomyopathy, in 36.7% of cases ischaemic cause was assessed, followed by tachymyopathy (25%), primary DCM (6.7%) and post myocarditis cardiomyopathy (5%). COPD (11.5%) and chronic kidney disease (CKD) (36.7%) were major comorbidities. At baseline, PFT demonstrated reduced DLCO, with a median of 60%, [IQR: 43.5-73], reduced FEV1 of 53% [IQR: 43-62], and reduced FVC of 57% [IQR: 50-63]. At discharge, FEV1 and FVC improved by 12.3% ±12% (p = 0.0014) and 14.8% ± 12.5% (p = 0.00042) (Figure 1). DLCO changed minimally (+1.3% ±14.4%; p = 0.71). Improvement of DLCO correlated with TAPSE at discharge (Beta = 0.32; 95% CI 0.06, 0.59, p = 0.023). Over a median follow-up of 2.2 years, all-cause mortality was 20.2% after three years. Baseline PFT measures were not associated with mortality.
Conclusions
In this AHF cohort, spirometric indices are markedly depressed but substantially recover with decongestion, while gas transfer remains largely unchanged. The link between DLCO improvement and right ventricular function (TAPSE) underscores cardiopulmonary coupling. Although underpowered for mortality endpoints, bedside PFT and DLCO offer pathophysiologic insights and may support management of hospitalised AHF patients.
Keywords
acute heart failure; pulmonary function; spirometry; DLCO; right ventricular function; decongestion
Figure 1
Changes in pulmonary function test measures from baseline to discharge.