Introduction: Imaging of Fluorine-19 labelled nanoparticles by magnetic resonance (19F MRI) offers a non-invasive approach for visualizing inflammatory processes in the myocardium following acute myocardial infarction (AMI). The 19F signal reflects the accumulation of labeled myeloid cells in the injured myocardial tissue. This accumulation is influenced by both the time of 19F nanoemulsion (NE) administration and NE dosage. In the present study, we employed 7T 19F MRI to monitor the temporal evolution of inflammation from the acute phase to resolution in a large animal model of AMI. By systematically varying the perfluoro-15-crown-5 ether (PFCE) dosage and timing, we aimed to optimize 19F deposition and enhance the visualization of inflammatory dynamics throughout the post-infarction period.
Method: AMI was induced in pigs by balloon occlusion of the coronary artery for 90 minutes. Intravenous administration of the PFCE was performed on days 2,4, and 6 post-AMI, using variable doses. In vivo 1H/19F MRI imaging was conducted on days 4-15 after AMI, followed by ex vivo after cardiac arrest on day 15.
Results and Discussion:
The acute phase of AMI is characterized by a pronounced infiltration of immune cells, primarily neutrophils and monocytes/macrophages, while the resolution phase is marked by a gradual decline in myeloid cell populations and a transition toward reparative immune activity. Various methods exist to assess and monitor immune response, and 19F MRI has gained attention for visualizing in vivo post-AMI inflammation owing its uptake by myeloid cells and negligible background. Here, we developed a robust 19F MRI approach to track post-AMI inflammation in large animal models at 7T by optimizing the timing and volume of PFCE injection. Our results showed that animals administered a 150 mL PFCE dose 2 days post-AMI had higher PFCE accumulation than at 4 days, indicating that early injection captured peak monocyte activity around day 4. This pattern is consistent with the findings of Temme et al. [1] in mice, showing that myeloid cells contribute to the 19F signal in injured myocardium during distinct phases of the immune response. Also, we administered 75 mL and 150 mL of PFCE 4 days after AMI to optimize dosage and assess myocardial accumulation. Although the 150 mL dose produced a stronger 19F signal, the 75 mL dose appeared sufficient for visualizing the injured myocardium while reducing overdose risk.
Conclusion: PFCE accumulation at the injury critically depends on both the timing and dose of administration. Early (2 days after AMI) administration of 150 ml PFCE yields optimal 19F signals, reflecting robust immune cell infiltration in the infarcted myocardium.
References
- Temme S, Jacoby C, Ding Z, Bönner F, Borg N, Schrader J, Flögel U. Technical advance: monitoring the trafficking of neutrophil granulocytes and monocytes during the course of tissue inflammation by noninvasive 19F MRI.
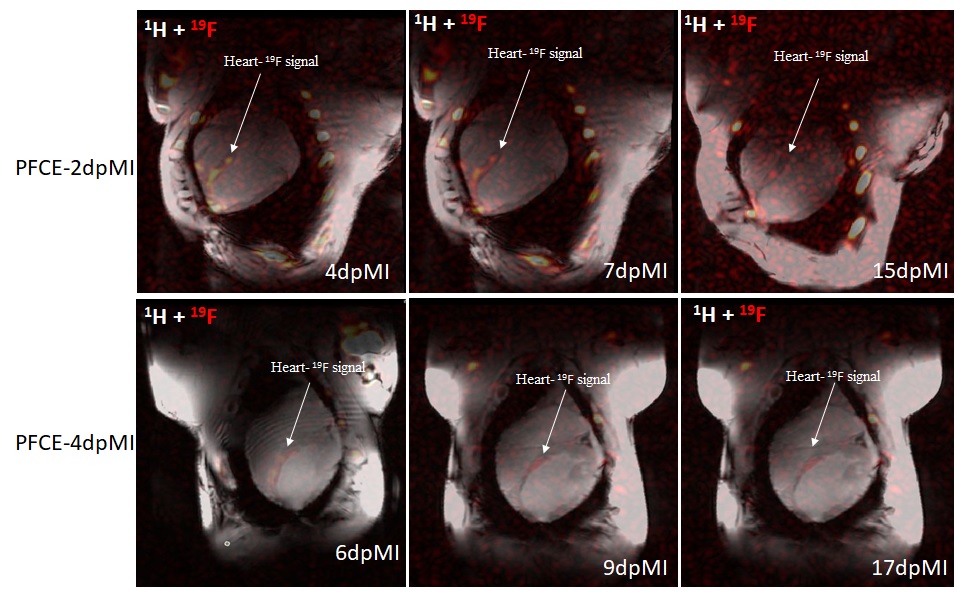
Figure 1: 19F MRI signal following PFCE administration. Injection at day 2 resulted in the highest signal, reflecting greater PFCE accumulation than 4 days.
