Background: The clinical benefit of additive ablation beyond pulmonary vein isolation (PVI) in atrial fibrillation (AF) remains uncertain. Nonetheless, substrate modification — particularly left atrial posterior wall isolation/ablation (LAPWa) — is frequently performed, mostly using pulsed field ablation (PFA) with large-footprint catheters. Although acute success rates are high, data on the long-term durability of LAPWa remain scarce.
Aim: To evaluate the rate of durable LAPWa during repeat ablation procedures following initial PFA-based LAPWa and to identify potential predictors of lesion durability.
Methods: All consecutive patients undergoing de-novo or repeat AF ablation including LAPWa using a penta-spline PFA catheter between January 2022 and July 2025 at one tertiary center were included. Baseline data, procedural characteristics and arrhythmia recurrences after a blanking period of 90 days prompting repeat ablation were assessed. Primarily reconnection of the LAPW in case of re-ablation was investigated. Furthermore, periprocedural safety and predictors of conduction recovery were analyzed.
Results: A total of 122 patients (70% male, n = 85/122; mean age 68,5 ± 9,2 years) were included. LAPWa was primarily performed in patients with persistent AF [107/122 (88%)] and redo procedures [111/122 (91%)]. A 3D mapping system was used in 86% (105/122), while 14% (17/122) were guided by fluoroscopy alone. The median procedure duration was 79 minutes (IQR 53-140 minutes), and fluoroscopy time 8.98 minutes (IQR 5,99-13,29 minutes). Periprocedural complications occurred in 9/122 (7%), including 1/122 (0.8%) major bleeding (arteriovenous fistula requiring surgery) and 2/122 (1.6%) minor bleedings (hematuria, AV fistula). Additional adverse events included 1/122 transient ischemic attack, and 1/122 stroke. No atrial-esophagus fistula (AEF) was observed.
After a median follow-up of 637 days (IQR 398-1020 days) 25/122 patients (21%) underwent repeat ablation for recurrent atrial arrhythmias. Durable LAPWa was confirmed in 14/25 (56%), while 11/25 (44%) demonstrated conduction recovery. 72% of patients underwent repeat catheter ablation due to recurrence of an atrial tachycardia (18/25). The most common AT mechanism was a perimitral macroreentry [7/18 (39%)], LAPW-dependent AT was observed in 1/18 (6%) patient.
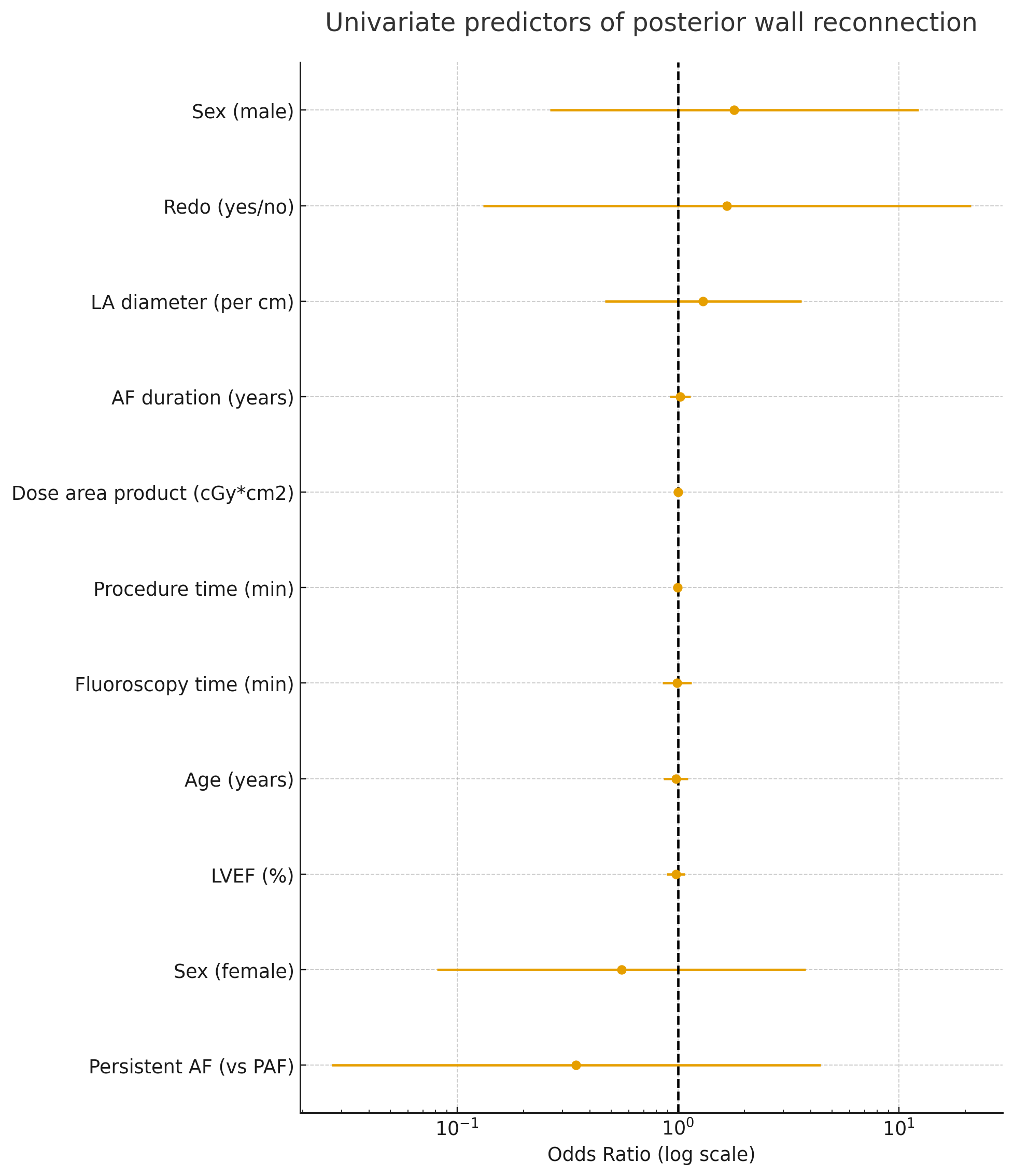
Univariate analysis revealed a non-significant trend for male gender larger left atrial diameter (OR 1.80 per centimeter increase; 95%-KI 0.26–12.30; p=0.549) being a potential predictor for recurrence. Similar trends were observed for redo procedures and larger left atrial diameter. Age, ejection fraction as well as AF duration were not associated with LAPW reconnection.
Conclusions: In this single-center observational study, LAPWa was durable in only 52% following PF ablation. Larger left atrial size, persistent AF, and multiple prior procedures may predispose to conduction recovery. Further studies with larger cohorts are warranted to optimize lesion durability and confirm these exploratory findings.