BackgroundPredicting neurological outcomes following cardiac arrest (CA) currently requires a resource-intensive multimodal assessment, including neurophysiology and brain imaging. The Better Rapid Assessment for In-hospital Neurological Outcome (BRAIN) study sought to develop a machine learning model to provide timely and accurate predictions using only readily available, routine in-hospital data.
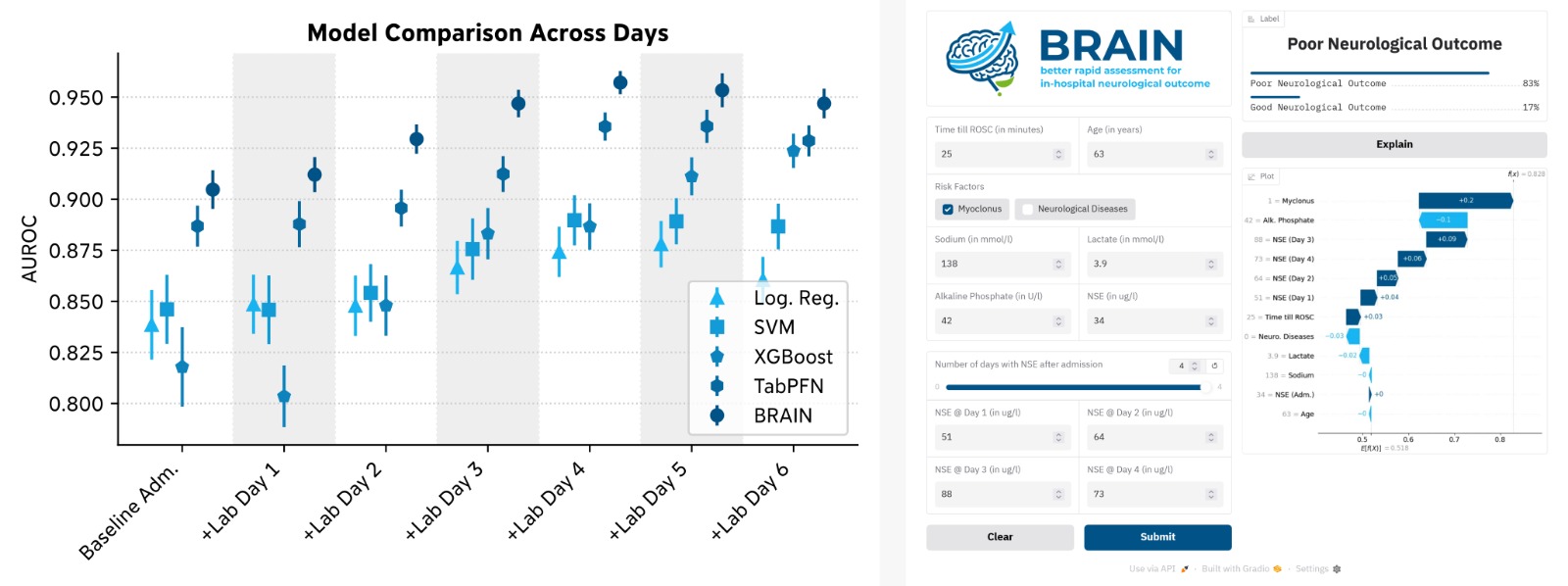
MethodsIn a cohort of 152 consecutive patients admitted to our cardiac intensive care unit after CA, unfavorable neurological outcome was defined as a Cerebral Performance Category ≥3. To predict unfavorable outcome, we evaluated several machine learning models, including logistic regression (LR), a support vector machine (SVM), extreme gradient boosting (XGBoost), and a tabular foundation model (TabPFN). Models were incrementally trained with baseline data, admission laboratory parameters, and daily laboratory parameters up to day 6. For the LR, SVM, and XGBoost models, missing data was imputed using nearest neighbors, classes were balanced, and hyperparameters were tuned. The best-performing model, termed BRAIN model, was then refined using feature selection based on feature importances derived from explainable artificial intelligence and clinical relevance. To improve accessibility, the final model was implemented as an interactive web calculator, available at https://hf.co/spaces/taltstidl/brain-score, which also visualizes individual feature importances, enhancing the interpretability of each prediction.
ResultsMedian age was 67 years, and 28% were women. An unfavorable neurological outcome occurred in 55% of patients. TabPFN achieved the highest area under the receiver operating characteristic curve (AUROC), rising from 0.88 at baseline to 0.94 at day 5 and plateauing thereafter. Peak AUROCs for LR, SVM, and XGBoost were 0.88, 0.89, and 0.92, respectively. Feature selection identified the most important predictors: age, preexisting neurological conditions, myoclonus at admission, time to return of spontaneous circulation, admission lactate, sodium, and alkaline phosphate, as well as daily neuron-specific enolase values. The BRAIN model using only this reduced feature set still achieved an AUROC of 0.90 at baseline, improving to a maximum of 0.96 at day 4.
ConclusionsThe BRAIN web calculator, which is based on a TabPFN machine learning model, offers accurate in-hospital prediction of neurological outcome after CA using only routinely available parameters, with continuous refinement during the first 6 hospital days. This tool may aid clinical decision-making in patients after CA.