Background
While energy-specific side effects involving hemolysis and renal function decline have been described for the pentaspline pulsed field ablation catheter (Penta-PFA), it remains to be addressed whether a novel balloon-in-basket PFA (BiB-PFA) catheter design with integrated contact sensing may mitigate these systemic effects. This study aimed to investigate differences in myocardial injury, inflammatory response, hemolysis and renal safety between the BiB-PFA and Penta-PFA catheter.
Methods
A total of n=40 consecutive patients with paroxysmal atrial fibrillation referred for de-novo pulmonary vein isolation (PVI) were prospectively included. PVI was performed in all patients using deep propofol sedation with either BiB-PFA (Volt with EnSite X integration, n=20) or Penta-PFA (Farapulse with fluoroscopy guidance, n=20). No additional ablation lesions were applied. Serial blood samples were collected before the procedure (T1), at the end of ablation (T2) and 24 hours after ablation (T3).
Results
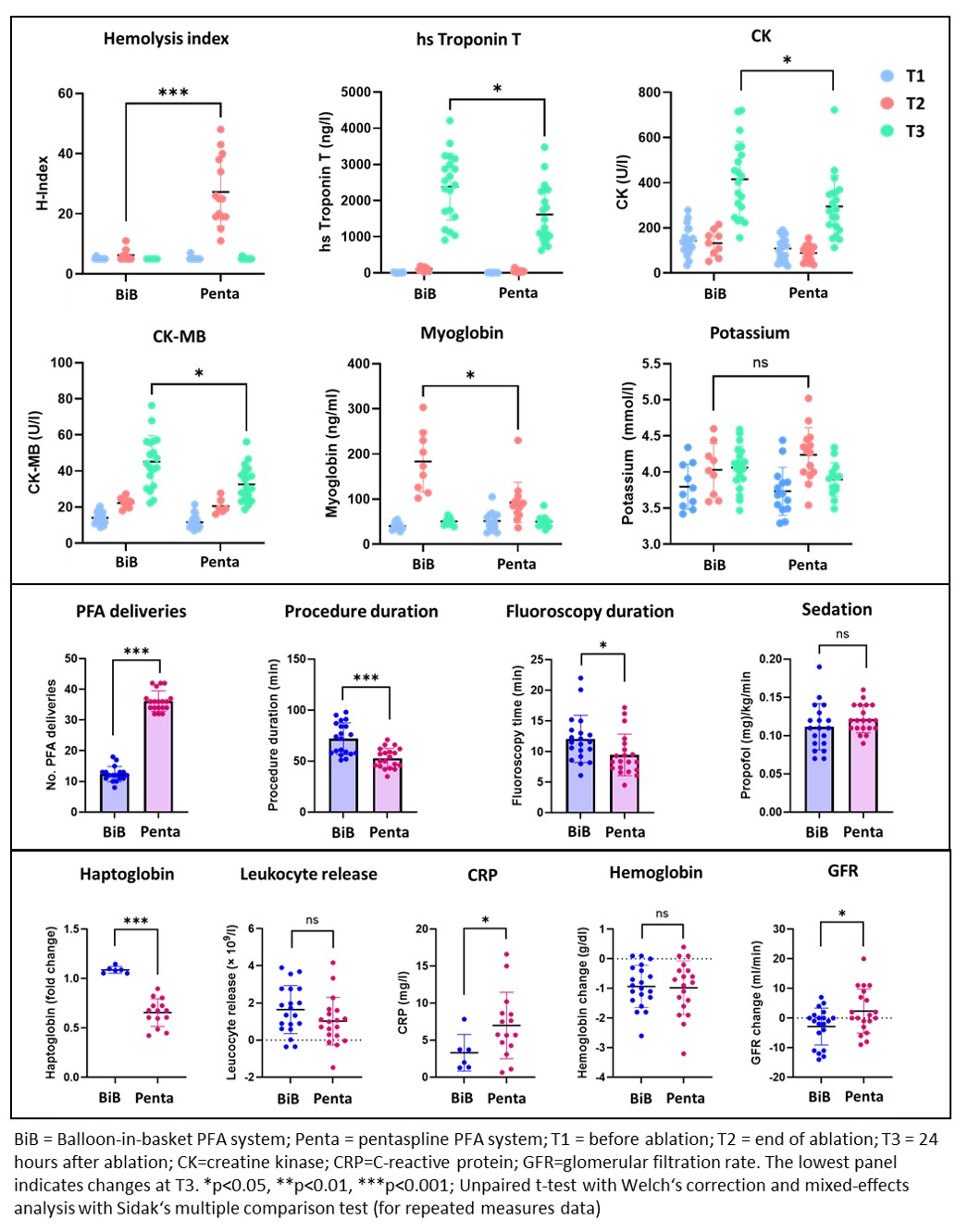
Baseline characteristics were well-balanced between the BiB-PFA and Penta-PFA group (age 62.3±10.3 vs. 66.5±10.1 years [p=0.201], female 25.0% vs. 50.0% [p=0.102]). PVI was achieved in all patients after a mean of 12.4±2.5 vs. 36.2±3.4 PFA deliveries (p<0.001). Procedure (72.0±15.6 vs. 53.0±9.8 min, p<0.001) and fluoroscopy duration (21.1±3.9 vs. 9.5±3.4 min, p=0.028) were longer with BiB-PFA. Propofol dosage was similar between groups (Figure 1). Hemolysis was not observed in the BiB-PFA group, while it was significant in Penta-PFA (Hemolysis index [T2] 5.9±2.3 vs. 27.3±11.4; haptoglobin fold change [T3] 1.1±0.1 vs. 0.7±0.2; both p<0.001). BiB-PFA was associated with a significantly higher release in high sensitivity troponin T (2383±928 vs. 1613±799 ng/l), CK (416±167 vs. 295±138 U/l, p=0.017), CK-MB (45±15 vs. 33±10 U/l, p=0.003) and myoglobin (183.6±67.7 vs. 92.6±44.6 ng/ml, p<0.001) and with lower inflammation (CRP 3.3±2.5 vs. 7.0±4.5 mg/l, p=0.029; Figure 1). While glomerular filtration rate dropped more significantly with BiB-PFA (-2.9±6.2 vs. 2.4±7.4 ml/min, p=0.021), no case of acute kidney injury occurred in any group.
Conclusions
The novel BiB-PFA system determines a more pronounced myocardial injury and lower inflammatory response without inducing relevant hemolysis. Whether the slight renal function decline is clinically relevant and related to the higher degree of myocardial injury with BiB-PFA warrants further investigation.