Background: With increasing prevalence of calcific aortic valve disease (CAVD) in aging populations, there is an urgent need for disease-modifying therapy beyond interventional or surgical approaches. Although atherosclerosis and CAVD share lipid accumulation and inflammation, LDL-lowering strategies fail to modify valvular calcification, suggesting cholesterol-independent drivers. Sphingolipids are potent mediators of metabolic inflammation and cell stress that may represent an unknown contributor to CAVD pathogenesis.
Purpose: This study aimed to investigate the pathogenic relevance of sphingolipid accumulation in CAVD, define underlying mechanisms and assess therapeutic efficacy of targeting sphingolipid synthesis in a reverse-translational approach.
Methods: We integrated lipidomics and proteomics of calcified human aortic valves with matched plasma to identify tissue- and disease-associated sphingolipid species and inflammatory markers. Their functional relevance was validated in a human valvular interstitial cell (hVIC) calcification model by stimulation with ceramides and precursors, and inhibition of key metabolic pathways. Calcification, osteogenic reprogramming, NF-κB-mediated inflammation, and ER stress were quantified by staining, enzyme assays, qPCR and protein analyses. Therapeutic efficacy was examined in ApoE⁻/⁻+PCSK9 AAVDY mice with metabolically induced CAVD using Myriocin, an inhibitor of sphingolipid de novo synthesis, administered in preventive and early-established disease stages. Valve structure and function, lipid composition and cell-state transitions were analyzed via echocardiography, histology, lipidomics and single-nucleus RNA sequencing (snRNA-seq).
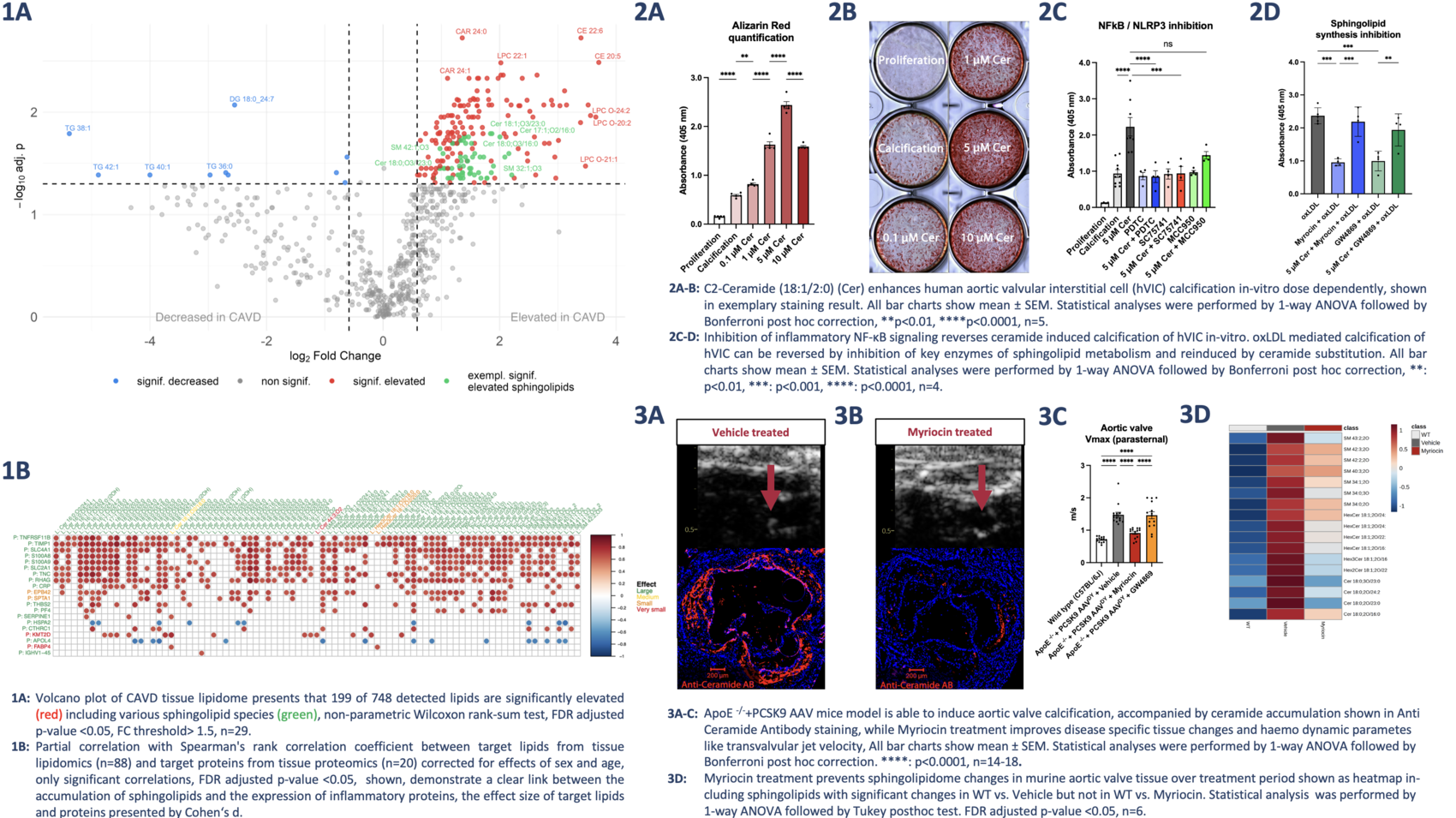
Results: Human calcified valves showed pronounced accumulation of long-chain ceramides (1A), correlating with NF-κB activation, endothelial-to-mesenchymal transition (EndMT), and osteogenic marker expression (1B). In vitro, ceramide and precursor stimulation induced hVIC calcification dose-dependently (2A-B). This effect was abolished by NF-κB inhibition, ER stress suppression, or blockade of sphingolipid synthesis (2C-D), underlining their functional relevance. In vivo, echocardiography revealed characteristic CAVD changes without treatment, e.g., increased valve thickness, while Myriocin improved cusp separation, Vmax and dPmean in preventive and early-disease settings, indicating functional valve rescue. Histologically, Myriocin reduced ceramide accumulation and mineralization, preserving valve morphology (3A-C). Long-chain ceramide species were conserved across species and reduced to near wild-type levels by Myriocin (3D). snRNA-seq showed reduced EndMT, decreased NF-κB signaling, attenuated ER stress and a shift of VIC composition toward quiescent phenotypes. Importantly, benefits occurred without lowering plasma LDL, indicating a cholesterol-independent mechanism.
Conclusion: Our research links sphingolipid-mediated lipotoxicity in calcified human valves via cross-species-validated signaling to the first proof-of-concept that modulation of sphingolipid metabolism mitigates CAVD progression in a cholesterol-independent manner, establishing the ceramide-inflammation axis as a promising target for CAVD therapy.