Background: Atrial fibrillation (AF) represents the most common sustained cardiac arrhythmia in adults and is linked to an increased risk of cardiovascular death. Specifically, AF is common across all etiologies of pulmonary hypertension (PH), including left ventricular (LV) disease, where right ventricular (RV) function is a critical determinant of prognosis. The AF burden has emerged as an important predictor of prognosis, particularly in patients with end-stage heart failure (HF). While there are some studies that have investigated the effects of AF on the LV, its effect on the RV remains largely unexplored.
Methods: Human cardiac slices from RV myocardium were freshly generated from patients with end-stage HF undergoing heart transplantation. Slices were long-term cultured and either stimulated regularly with 60 bpm (control) or with an AF-simulating arrhythmia burden of 30% and 70% at 90 bpm (low/high AF burden). Further, experiments were performed using RV human-induced Pluripotent-Stem-Cell-Cardiomyocytes (RV-hiPSC-CMs). After 48 hours and 7 days, epifluorescence microscopy and immunofluorescence staining for α-actinin and titin were performed to assess calcium homeostasis and sarcomere regularity.
Results: A total of 35 RV cardiac slices from 5 donors with end-stage HF were investigated. After 2 days, the AF burden groups already showed a significantly reduced twitch amplitude compared to the control. This effect remained consistent over time until day 7, especially in the high AF burden group (p=0.004), pointing to contractile dysfunction due to arrhythmic stimulation. No significant differences were observed in diastolic tension, relaxation time, time-to-peak, post-rest ratio and forced-frequency relationship.
In RV-hiPSC-CM, after 48 hours (n=79/5) and 7 days (n=80/6), calcium transient amplitude was significantly reduced in the group with high AF burden. After 7 days, sarcomere organization measured by α-actinin (n=233/7) and titin (n=237/7) regularity was significantly impaired in the group with high AF burden compared to the control and low AF burden group (p<0.001).
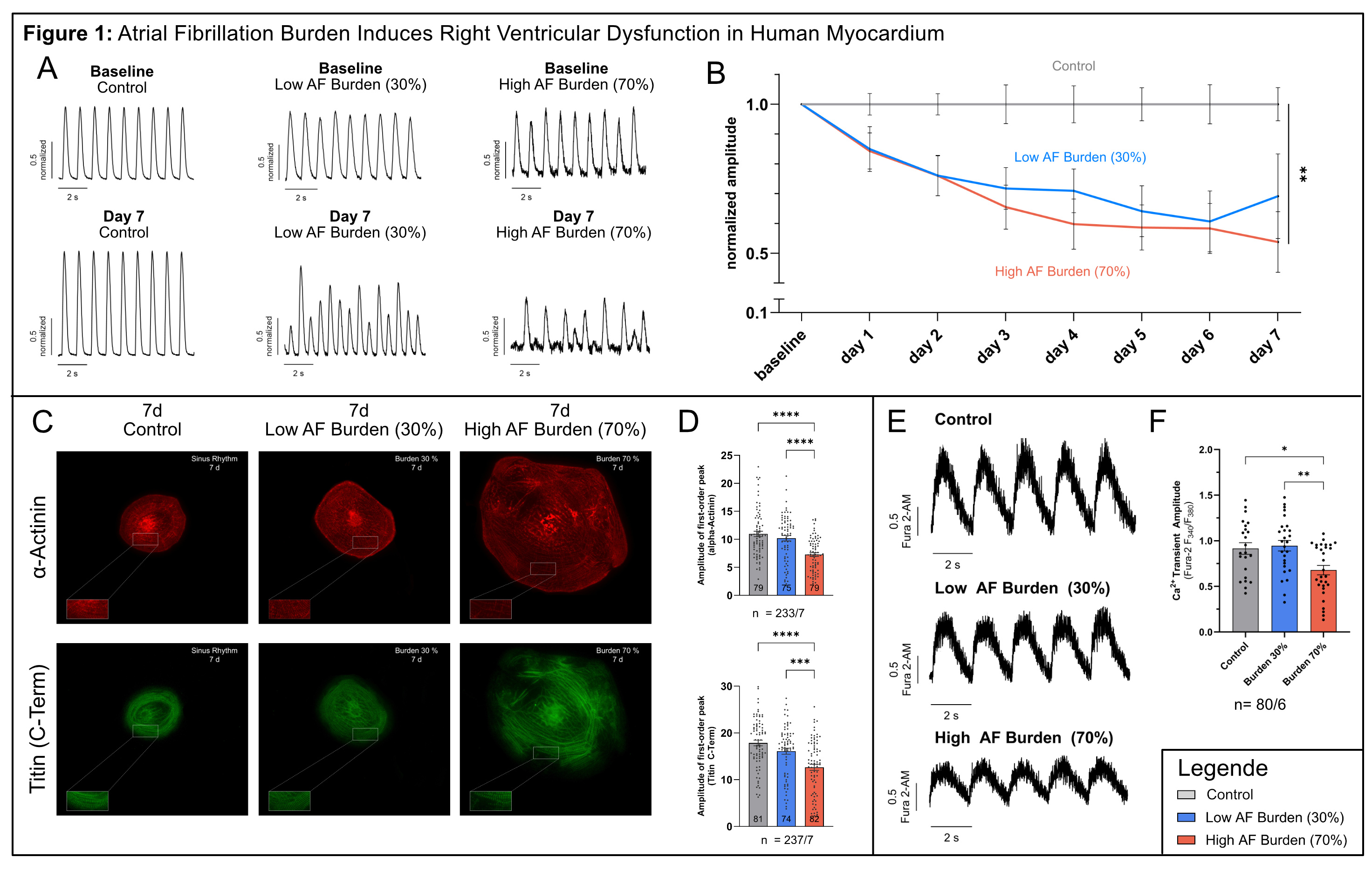
Fig. 1: A Original traces of control, low and high AF burden RV cardiac slices (day 0 and 7). B Normalized twitch amplitudes during AF stimulation. C Original pictures of stained RV-hiPSC-CM for α-actinin and titin. D Results of sarcomere regularity analysis. E Representative calcium transients of control, low and high AF burden RV-hiPSC-CMs (7d). F Calcium transient amplitude after 7d stimulation. Data as mean ± SEM (*p≤0.05, **p≤0.01, ***p≤0.001, ****p≤0.0001).
Conclusion: We observed a reduction in contractile force in RV slices exposed to AF burden simulation, which could be explained by impaired calcium handling and sarcomere disorganization in an RV-hiPSC-CM model. These findings provide new insights into the interplay of AF and HF. Further experiments will explore the underlying mechanisms and assess the effects of in vitro rhythm control simulation in RV human myocardium. Moreover, further studies aim to clarify the dual burden of AF and PH on RV function.
