Background: Chronic inflammation is frequently observed in heart failure (HF), and elevated inflammatory markers are associated with incident HF. The direct impact of persistent inflammation on cardiac structure and function remains incompletely understood.
Aim: To evaluate the influence of chronic, stable inflammation on left-ventricular structure and function.
Methods and results:
We quantified hsCRP by immunonephelometry in patients admitted to the Department of Internal Medicine I at UKA between June 2018 and June 2019. Inclusion required a stable inflammatory status, defined as two independent hsCRP measurements obtained ≥24 hours apart falling within predefined limits. Of 114 screened individuals, 77 met the stability criterion and were subsequently stratified into three hsCRP categories: low (0.0–0.9 mg/L), medium (1.0–2.9 mg/L), and high (3.0–9.9 mg/L).HF (HFrEF, HFmrEF, HFpEF) was present in 80% of the cohort. Echocardiography assessed LV ejection fraction (EF), global longitudinal strain (GLS), and diastolic parameters (E, A, septal/lateral e’). HF subtype distribution did not differ across hsCRP groups (p > 0.05). Patients in the highest hsCRP group showed significantly reduced early diastolic LV relaxation, reflected by lower septal and lateral e’ compared with the low-hsCRP group (mean e’: 6.7 ± 1.9 vs. 5.4 ± 1.1; p = 0.03), independent of LVEF. Subgroup analysis revealed that this association was specific to HFpEF (Rho = –0.50; p = 0.022), with no significant correlations in HFrEF/HFmrEF or non-HF patients. Similarly, impaired GLS correlated with inflammation only in HFpEF (Rho = 0.60; p = 0.008).
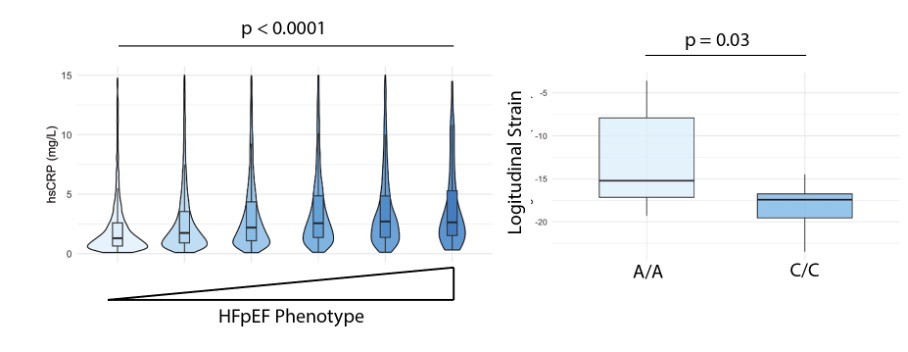
To validate these findings, we extended our analysis to the UK Biobank. At baseline, 15,853 participants had a diagnosis of HF. The HFpEF-associated clinical profile—older age, hypertension, atrial fibrillation, female sex, and obesity—showed a strong association with higher hsCRP levels in HF patients (Figure 1). To further investigate the role of inflammation, we additionally evaluated the anti-inflammatory IL-6–modulating SNP rs2228145 (A>C) in HF patients with an HFpEF phenotype. In an exploratory analysis, homozygosity for the C-allele was associated with significantly better longitudinal strain compared with A/A carriers (A/A: –13.1 ± 5.9 vs. C/C: –18.2 ± 2.8; p = 0.03), supporting a potential causal role of inflammatory pathways in functional LV impairment in HFpEF (Figure 1).
Conclusion:
Chronic low-grade inflammation is associated with impaired myocardial relaxation and deformation in patients with a HFpEF phenotype, suggesting a pathophysiological link between inflammatory activity and early LV dysfunction in this subgroup.
Figure 1: (A) Inflammation in HF patients according to a HFpEF clinical profile (B) Logitudinal Strain in HF patients with impaired IL6-signalling (C/C).