Background: ATTR amyloidosis is a systemic disease caused by misfolded transthyretin proteins aggregating in various tissues. In case of cardiac involvement, most patients develop a progressive and restrictive cardiomyopathy (CM) with global myocardial hypertrophy affecting both ventricles. Early prognostic markers are essential for optimal patient management and personalized consultation against the background of increasing diagnostic and therapeutic options and rising patient numbers. The aim of the present analysis is to identify predictors of prognosis from clinical routine data of patients with ATTR-CM and to determine whether these predictors change in ATTR-specific therapy cohort.
Methods: The analysis includes data from 140 patients with confirmed ATTR-CM of the patient registry of the Amyloidosis center of lower Saxony obtained between 04/2019 – 10/2025. Clinical data encompasses medical history, medication, electrocardiogram, laboratory results and echocardiography. The primary endpoint death was collected through once yearly during follow-up. Variables with missing data of <10% were analysed; missing values were imputed using the mean and mode. We analysed binary variables using chi-square test, while continuous variables were tested for normal distribution with Shapiro-Wilk test and analysed using Student's t-test or Mann-Whitney U test. Associations of variables with mortality were analysed using univariate and multivariate logistic regression.
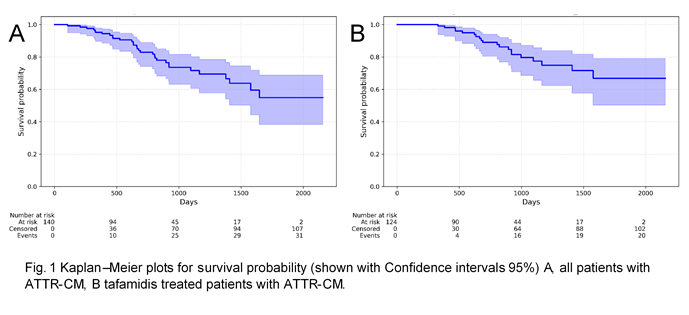
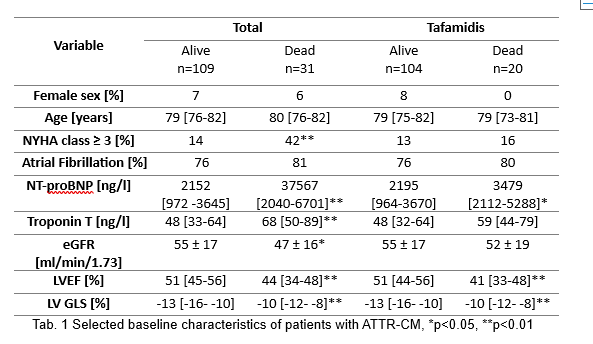
Results: A total of 140 patients with confirmed ATTR-CM (129 wild-type, 11 hereditary) were analysed. Of these, 124 were treated with tafamidis. The median follow-up time was 896 days (total) and 924 days (tafamidis). 31 patients (22%) in the total cohort and 20 (16%) in the tafamidis cohort died during follow-up (Fig. 1). Between the groups dead or alive, 24 and 11 of 86 clinical variables at baseline showed a significant difference in the total and tafamidis cohort, respectively. In univariate logistic regression-analysis 19 (total) and 11 (tafamidis) variables were significantly associated with all-cause death. After performing multivariate logistic regression analyses troponin T (p=0.01) and the echocardiographic parameter, left ventricular outflow tract velocity time integral (LVOT VTI) (p=0.02) remained independent predictors of mortality in the total cohort. In the tafamidis cohort, only the echocardiographic parameters TAPSE and LVOT VTI remained independently associated with all-cause death.
Conclusion: Predictors of death obtained from clinical routine data seem to change with tafamidis treatment in patients with ATTR-CM. With tafamidis treatment in particular parameters reflecting right and left ventricular function seem to be important predictors for prognosis and may be important indicators of disease progression.