Background: Left ventricular systolic dysfunction (LVSD) frequently occurs in patients with atrial tachyarrhythmia (TAA). While short-term improvement of left ventricular (LV) function after rhythm control therapy has been demonstrated, long-term structural recovery and its association with myocardial fibrosis remain insufficiently characterized.
Methods: Fifty patients with TAA (heart rate >100 bpm) and newly diagnosed, otherwise unexplained LVSD (mean age 68.1±11.2 years, baseline LV ejection fraction (LVEF) 35.7±8.6%) who underwent successful rhythm control treatment were prospectively followed up. Survival status of all patients was obtained during long-term follow-up (mean follow-up duration 4.7 years). In addition, paired cardiac magnetic resonance imaging (cMRI) at baseline and long-term follow-up was available in 22 patients (mean interval between cMRIs 3.4 years). LV function, geometry and late gadolinium enhancement (LGE) were assessed. Further, complete LV reverse remodeling (CRR) defined as an LVEF increase ≥10 U and a LV end-diastolic volume index (LVEDVi) decrease ≥10% was evaluated. Patients who met only one of these criteria were classified as having partial reverse remodeling (PRR).
Results: In total, 11 of the 50 baseline patients (22%) died during long-term follow-up. Compared to survivors, non-survivors were older (77.5±9.3 years vs. 65.4±10.3 years, p=0.001), presented with a higher initial heart rate (137.6±17.9 bpm vs. 122.6±13.8 bpm, p=0.005) and had a higher amount of LGE in the first cMRI (12.0±5.8% vs. 7.4±4.6%, p=0.036). ROC analysis identified an LGE threshold of 12% as the optimal cut-off to discriminate survivors from non-survivors. Patients with higher LGE (≥12%) showed significantly lower long-term survival compared to those with lower LGE (<12%) (p=0.014, figure 1).
At baseline, patients with paired cMRIs had a mean age of 63.3±10.8 and 73% were male. During long-term follow-up, LVEF increased from 36.1±12.7% to 46.8±11.3% (p=0.005), while LV end-diastolic diameter decreased from 61.0±6.7 mm to 55.2±9.2 mm (p=0.0003), LV end-systolic diameter from 49.8±10.2 mm to 43.5±11.3 mm (p=0.001), LVEDVi from 104.1±30.8 mL/m2 to 92.2±30.3 mL/m2 (p=0.02) and LV end-systolic volume index from 68.8±29.0 mL/m2 to 50.8±24.6 mL/m2 (p=0.002). LGE extent remained unchanged (6.5±4.3% vs 6.4±4.5%, p=0.329). Twelve of 22 patients (55%) showed CRR, while 9 showed PRR (41%) and 1 patient (5%) did not fulfill any remodeling criteria.
Conclusion: In this cohort of rhythm-controlled patients with TAA and LVSD, sustained functional and structural reverse remodeling of the LV was observed, although many patients only showed PRR. The amount of baseline LGE was associated with long-term mortality, highlighting its prognostic significance in this population. These results emphasize the value of cMRI for long-term risk stratification in patients with TAA and LVSD.
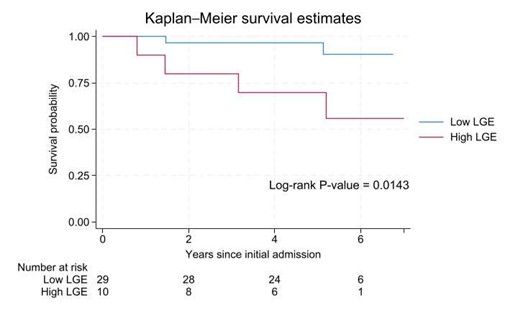
Figure 1: Kaplan–Meier survival curves according to baseline LGE.
