Background Recurrent atrial fibrillation (AF) after pulmonary vein isolation (PVI) remains a therapeutic challenge, primarily driven by pulmonary vein (PV) reconnection. Pulsed field ablation (PFA) has emerged as a novel, non-thermal energy source with high myocardial selectivity and favorable safety profile. However, data on its use for repeat AF ablation, particularly in conjunction with 3D electroanatomic mapping, are limited.
Aims This study aimed to evaluate the feasibility, efficacy, and safety of 3D-mapping guided PFA for repeat PVI in patients with recurrent AF.
Methods Patients referred for repeat AF ablation after prior PVI were prospectively enrolled. Exclusion criteria included prior 3D-guided LA ablation with known anterior or septal low-voltage areas or documented atrial tachycardia recurrence (except typical cavotricuspid isthmus (CTI)-dependent flutter). All procedures were performed using a commercially available PFA system under 3D electroanatomic guidance. The pentaspline ablation catheter served as both mapping and ablation tool and no dedicated mapping device was utilized. Baseline and post-ablation 3D LA voltage maps were obtained in all cases. Additional substrate ablation (posterior wall or roof isolation) was performed per institutional standard if low-voltage areas were identified; anterior ablation was limited to cases with inducible atrial tachycardia involving the septum. Procedural parameters, PV reconnection rates, substrate localization, and acute outcomes were analyzed.
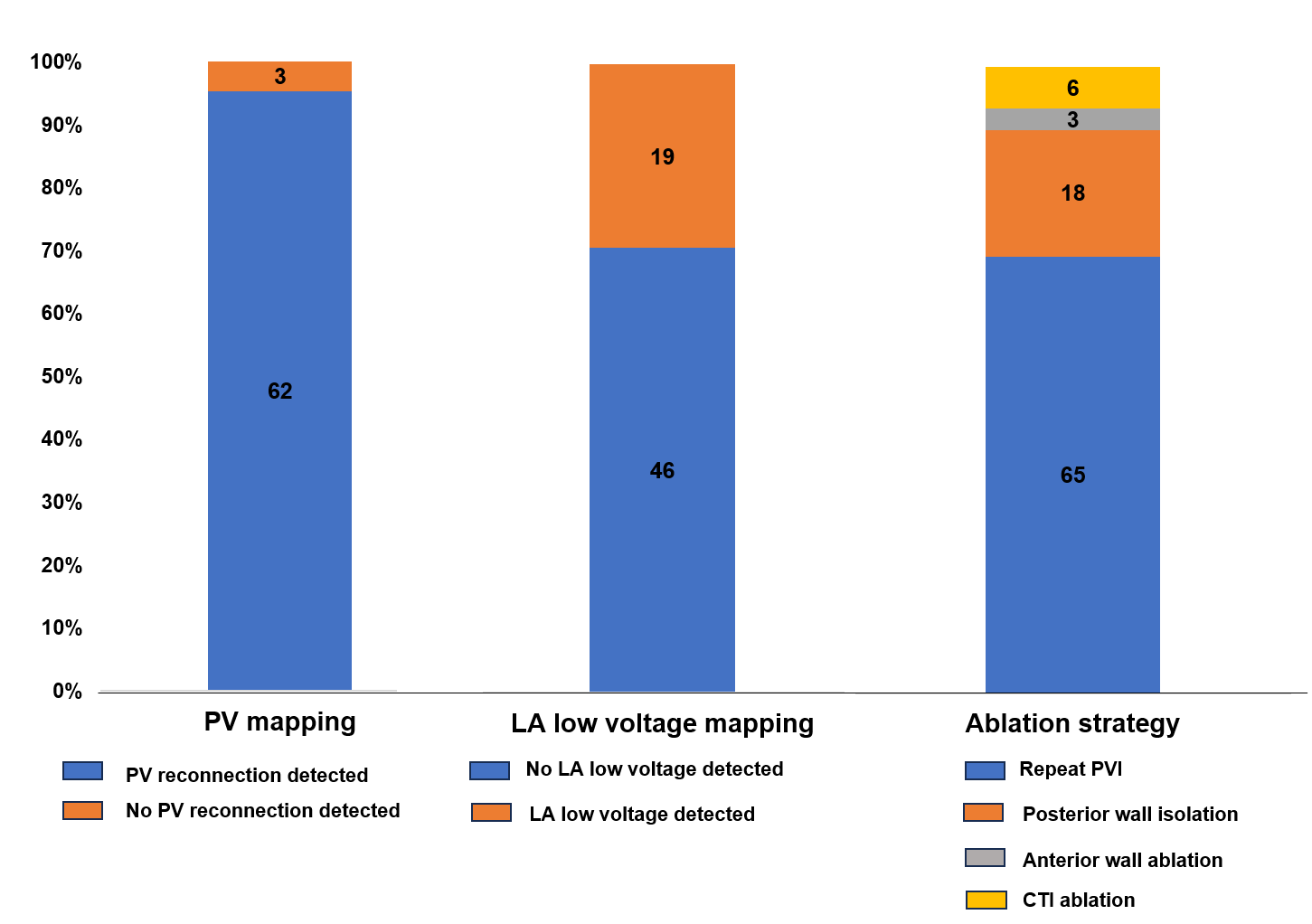
Results A total of 65 patients (mean age 65.6±11.4 years; 84.6% male) underwent 3D-mapping guided PFA for repeat PVI. Prior ablation had been performed with radiofrequency energy in 35 (53.8%), cryoballoon devices in 22 (33.9%), and PFA in 2 (3.1%) patients. PV reconnection was identified in 62 patients (95.4%). Low-voltage areas were detected in 19 patients (29.2%), primarily at the posterior wall (n = 15, 88.2%), but also in the anterior LA (n = 4, 23.5%) and roof (n = 2, 11.8%). Additional ablation beyond PVI was performed in 25 patients (38.5%), including posterior wall isolation in 18 (69.2%), anterior wall ablation in 3 (11.5%), and CTI ablation in 6 (23.1%). Figure 1 summarizes intraprocedural findings. The mean total number of PFA applications was 36.6±11.2 and mean procedure duration was 51.6±18.2 minutes. All procedures were successfully completed without major complications.
Conclusions 3D-mapping guided pulsed field ablation for repeat PVI is feasible allowing visualization of PV reconnection and low voltage areas, enabling tailored ablation strategies while maintaining short procedure times and an excellent safety profile. These findings support the integration of 3D-mapping with PFA technology for repeat AF ablation procedures.