Background:
Resident cardiac macrophages couple to cardiomyocytes of the cardiac conduction system through connexin-43–type gap junctions, thereby modulating the action potential of atrioventricular nodal cells and impacting impulse propagation. We hypothesized that cardiomyocyte-macrophage coupling is bidirectional, i.e. that cardiac electrical activity may modulate the activity and secretory profile of resident immune cells in return. To test this hypothesis, we measured intracardiac chemokine and cytokine responses to controlled electrical pacing during routine electrophysiological studies.
Methods:
72 patients undergoing clinically indicated electrophysiological studies for supraventricular tachycardia evaluation were prospectively enrolled. Paired blood samples were collected both peripherally and intracardially, via a multipurpose catheter positioned in the coronary vein sinus, both before and immediately after intracardiac electrical pacing and prior to ablation, if performed. Concentrations of 22 serum factors with established macrophage relevance were quantified using multiplex fluorescence immunoassay and ELISA. Pre- and post-stimulation values were compared for intracardiac and peripheral compartments, and gradients were calculated to assess local cardiac release.
Results:
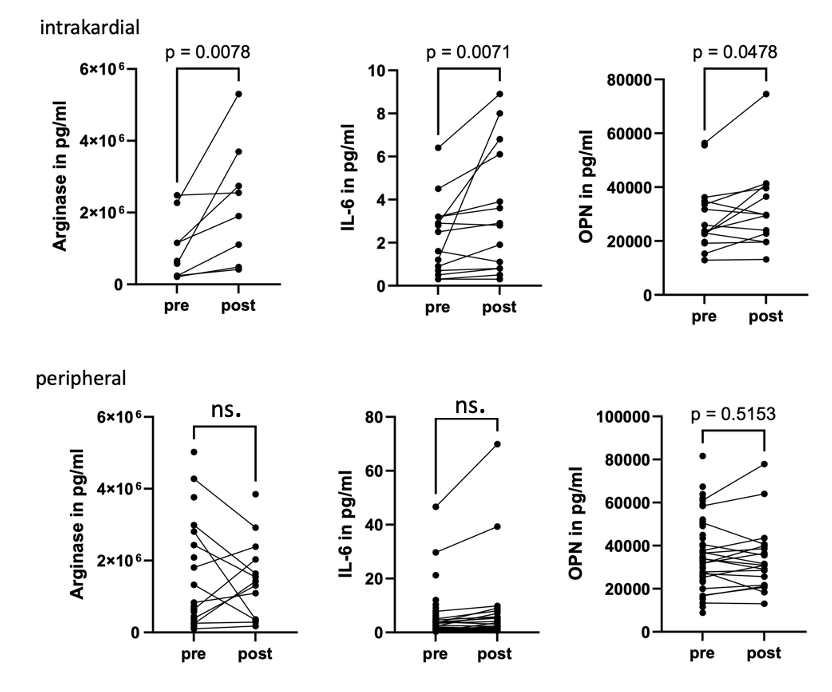
Electrical stimulation induced significant intracardiac increases in Arginase, IL6 and Osteopontin compared to baseline in normofrequent patients, indicating release is independent of chronic tachycardia pathology. Arginase demonstrated a distinct cardiac-periphery gradient, indicating a local intracardiac source. In peripheral venous samples, IL-6, CXCL9 and CXCL10 levels rose significantly after pacing.
Conclusions:
Although it remains to be determined if acute peripheral increases are due to cardiac source spillover or extracardial release, locally restricted release of arginase points to an immediate immune response within the cardiac microenvironment triggered by electrical excitation, even in the absence of structural myocardial injury. Our in vivo evidence that cardiac electrical stimulation elicits acute immune factor response, some of them systemic and some locally restricted, is in line with the hypothesis of gap-junction mediated signalling from cardiac myocytes to resident macrophages. The observed release of IL-6, osteopontin, arginase-1, CXCL9, and CXCL10 uncovers a potential mechanistic link between acute electrical activity and immune modulation in the heart.