Background:
Although myocardial fibrosis (MF) is common in patients with aortic stenosis (AS) and independently predicts adverse outcomes, cardiac MRI (CMR) is not routinely available in those with moderate AS (mAS) to assess MF. Circulating biomarkers of extracellular matrix remodeling, including TIMP-1, Gal-3, and sST2, have been linked to MF and cardiac inflammation. We investigated whether plasma biomarkers reflect CMR-derived indices of MF and inflammation in patients with mAS.
Methods:
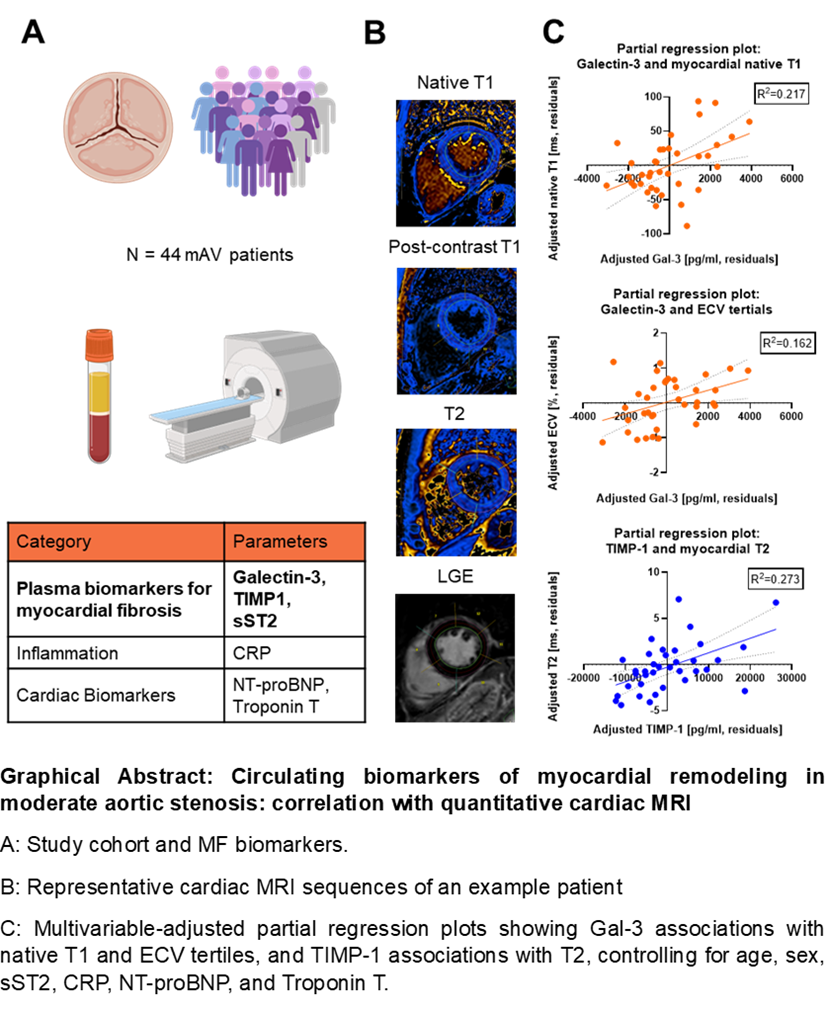
In this prospective study, 44 patients (82% men; mean age 76.6 ± 7.5 years) with mAS underwent 1.5 T CMR between December 2023 and October 2025. Same-day plasma samples were analyzed for MF biomarkers by ELISA (Fig.1A). Associations with CMR markers of fibrosis and inflammation were evaluated using linear regression and compared with established markers of inflammation, cardiac injury, and remodeling. Patients were further stratified into tertiles of native T1, extracellular volume (ECV), T2 and extent of late gadolinium enhancement (LGE) to identify patient-based differences in CMR-based fibrosis and inflammation patterns (Fig.1B).
Results:
Multivariable regression analyses revealed distinct associations between circulating biomarkers and quantitative CMR indices. For native T1, Gal-3 showed the strongest association in multivariable regression analyses, adjusted for age, sex, other MF biomarkers, CRP, NT-proBNP and troponin T (β=0.485, p=0.009) (Fig. 1C) ECV tertiles were independently associated with adjusted Gal-3 (β=0.405, p=0.030) (Fig.1C), with the highest ECV tertile (>29.3%) linked to elevated troponin T, NT-proBNP, and reduced LVEF, indicating subclinical myocardial dysfunction. T2 was most strongly associated with adjusted TIMP-1 (β=0.558, p=0.003) (Fig.1C). In univariable analyses, LGE extent correlated with TIMP-1 (Pearson r=0.504, p=0.001), NT-proBNP (Pearson r=0.451, p=0.004), and troponin T (Spearman r=0.465, p=0.002). LV mass was associated solely with adjusted sST2 (β=-0.496, p=0.013). Clinical characteristics, including NYHA class and comorbidities, did not differ significantly across CMR marker tertiles.
Conclusions:
In mAS, circulating biomarkers of extracellular matrix turnover and inflammation, particularly TIMP-1 and Gal-3, showed distinct and independent associations with CMR indices of myocardial fibrosis and inflammation after age, sex and biomarker adjustments. Gal-3 was most strongly associated with higher native T1 and ECV values, consistent with diffuse interstitial fibrosis, whereas TIMP-1 was primarily linked to elevated T2 and LGE, reflecting tissue inflammation and focal fibrosis. These findings suggest that Gal-3 and TIMP-1 may capture complementary aspects of the myocardial fibrotic–inflammatory continuum in mAS. Together, they support the potential utility of plasma biomarkers as accessible adjuncts to CMR for early myocardial characterization, warranting validation in larger, outcome-driven cohorts.