Background: The adoption of transcatheter mitral valve replacement (TMVR) in clinical practice has been limited by high screening failure rates, largely due to anatomical factors like the risk for neo-left ventricular outflow tract (neo-LVOT) obstruction. A novel transseptal TMVR system employing a unique left atrial (LA) fixation mechanism has recently been introduced, offering the potential to improve anatomical eligibility without affecting the LVOT.
Aim: This study aimed to evaluate anatomical eligibility for a transseptal TMVR system with LA fixation in patients with relevant mitral regurgitation (MR) previously screened for TMVR, and to assess the potential to improve overall TMVR eligibility.
Methods: All consecutive patients with relevant MR referred for TMVR evaluation were prospectively enrolled in a single-center TMVR screening registry. For this study, all patients that underwent screening for dedicated TMVR devices and had full cardiac cycle CT imaging were included. These patients underwent retrospective eligibility assessment using manufacturer-recommended CT measurements for the novel LA fixation TMVR system. Based on the screening outcomes, patients were categorized into either a Screen Pass or Screen Failure group. Reasons for ineligibility were analyzed, and clinical and CT parameters were compared between groups. The potential impact on overall TMVR eligibility was assessed comparing the real-world acceptance rate (including only sub-valvular TMVR devices) versus the expected screen pass rate using the LA fixation TMVR device.
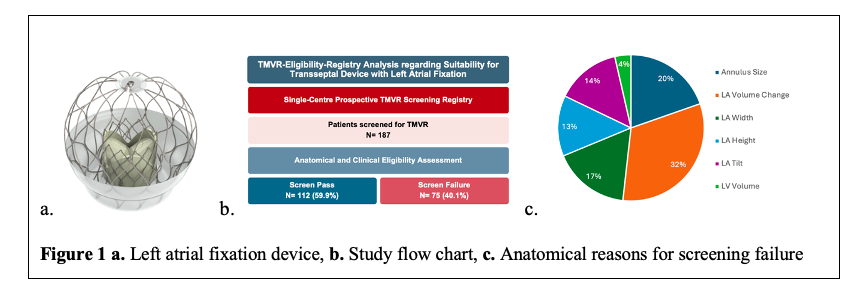
Results: A total of 211 patients underwent TMVR screening. Of these, 24 patients were excluded due to missing CT imaging, and 187 were included in the analysis (mean age 76.3 years [IQR 73-81], 55.6% male, ejection fraction 45.8% [IQR 34-57], 48.9% primary MR). Following CT-based anatomical evaluation, 112 patients (59.9%) met the eligibility criteria (Screen Pass), while 75 patients (40.1%) did not (Screen Failure). The most common reasons for screening failure were high LA volume change (N=36, 48.0% of Screen Failure patients), mitral annular size (N=22, 29.3%) and LA width (N=19, 25.3%). Patients in the Screen Pass group were more frequently male (57.1% vs. 53.3%) and had a higher prevalence of atrial fibrillation (82.14% vs. 57.33%). The predominant MR etiology was secondary in the Screen Pass group (N=72, 66.06%), whereas most patients in the Screen Failure group had primary MR (N=43, 58.90%). Comparison of the real-world acceptance rate for screened sub-valvular TMVR devices (33.6%) with the expected screen pass rate using the novel LA fixation TMVR system (59.9%) suggested a potential improvement of overall TMVR eligibility by 26.3%.
Conclusion: The inclusion of this novel transseptal TMVR device with LA fixation to the TMVR screening portfolio has the potential to expand overall TMVR suitability. While the risk of neoLVOT obstruction remains a major limitation for many sub-valvular TMVR devices, this novel device completely spares the LVOT. Further clinical studies are needed to determine whether TMVR treatment of eligible patients can provide prognostic benefit in patients with severe MR.