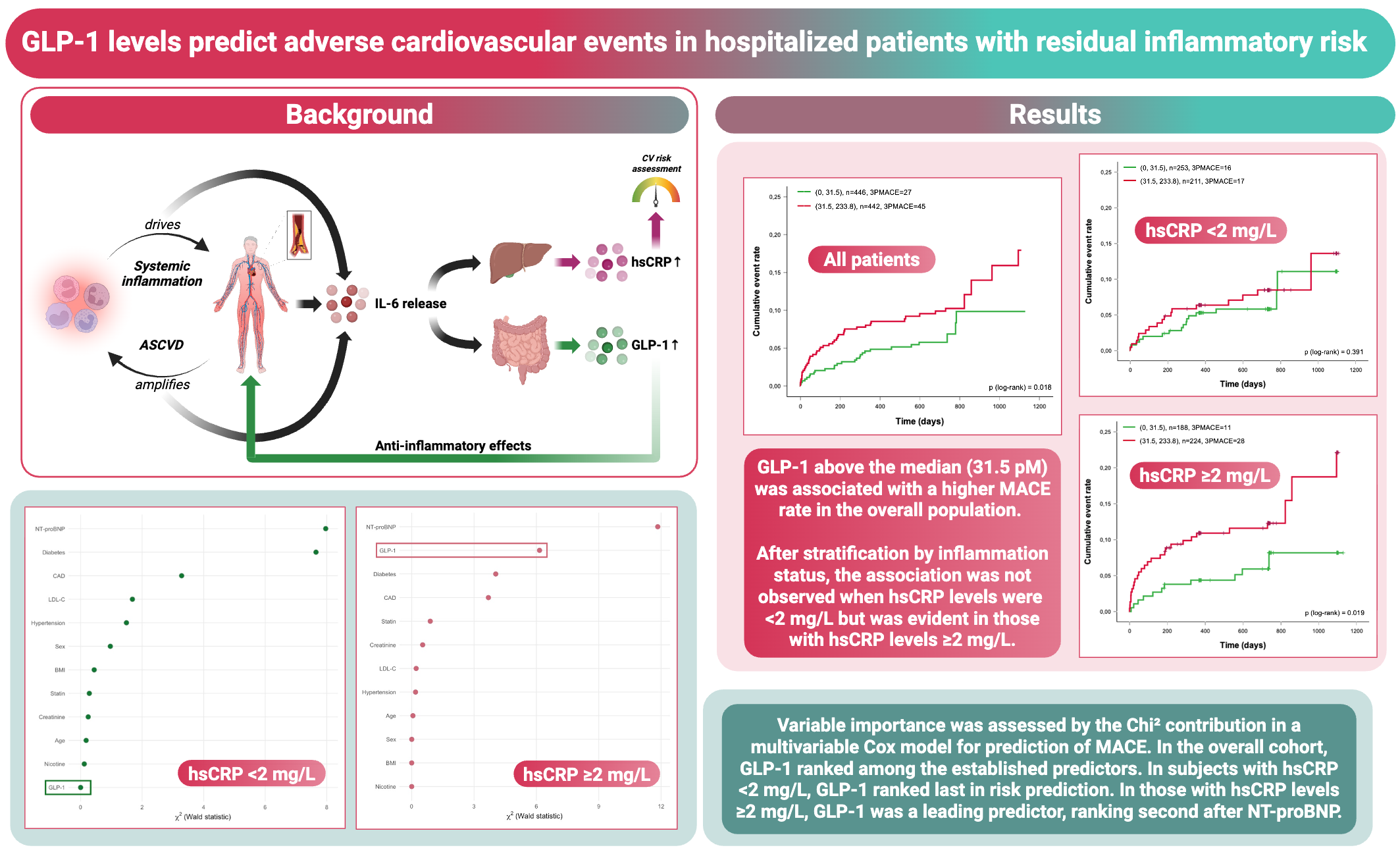
Background: Glucagon-like peptide-1 (GLP-1) is a gut-derived incretin hormone secreted in response to food-intake and pro-inflammatory stimuli. Secretion of GLP-1 leads to post-prandial insulin secretion and subsequent glucose lowering. Beyond metabolic effects, GLP-1 has pleiotropic and cardioprotective effects and GLP-1 receptor agonists reduce cardiovascular (CV) events in patients with diabetes or obesity. Direct anti-inflammatory effects are likely to contribute to beneficial effects of GLP-1RA. Clarifying the prognostic relevance of endogenous GLP-1 levels in CV disease could improve our understanding of how inflammation interacts with incretin pathways.
Methods: Circulating GLP-1 levels were measured in 888 fasted hospitalized patients in a cardiology department within a prospective single-center biobank study. The primary endpoint was a composite of non-fatal myocardial infarction, non-fatal stroke and CV death (MACE: major adverse cardiovascular events). Analyses were performed in the full cohort and stratified after individuals with and without residual inflammatory risk, defined by high-sensitivity C-reactive protein (hsCRP) levels above 2 mg/L. The association of GLP-1 with MACE was assessed using Kaplan-Meier curve, multivariable Cox proportional hazards and variable importance analyses.
Results: Median age of the cohort was 70 years, 65% were male and median GLP-1 levels were 31.5 pM. During a median follow-up period of two years, MACE was observed in 72 out of 888 patients. In Kaplan Meier curve analyses, GLP-1 levels above the median were associated with a higher rate of MACE (log-rank p=0.018). In univariable analyses, higher GLP-1 concentrations were associated with increased risk of MACE (GLP-1 above median: Hazard ratio (HR): 1.77; 95% confidence interval: 1.10, 2.85; p=0.019). These results remained significant after multivariable adjustment for age, sex, BMI, smoking, hypertension, type 2 diabetes, statin use, coronary artery disease, creatinine, LDL cholesterol and NT-proBNP (p=0.026). In variable importance analyses assessing the individual contribution of each variable from the multivariable model to prediction of MACE, GLP-1 ranked among the top predictors of outcome, outperforming traditional CV risk markers such as LDL cholesterol and creatinine. Importantly, in patients without systemic low-grade inflammation (hsCRP <2 mg/L), GLP-1 was not associated with MACE (log-rank p=0.391; HR: 1.39; 95% CI: 0.68, 2.67, p=0.393) and ranked last in variable importance analyses. Whereas in patients with hsCRP levels ≥2 mg/L, higher GLP-1 levels were strongly associated with MACE (log-rank p=0.019; HR: 2.25; 95% CI: 1.12, 4.52; p=0.023) and showed high variable importance, ranking second after NT-proBNP.
Conclusions: In hospitalized stable patients with CVD elevated circulating GLP-1 levels independently predicted MACE, with the strongest associations in those with residual inflammatory risk. These findings support the hypothesis of GLP-1 as an endogenous counter-regulatory peptide that rises in response to inflammatory activation. GLP-1 may serve as a marker of residual inflammatory risk, representing a compensatory, anti-inflammatory mechanism.