Background
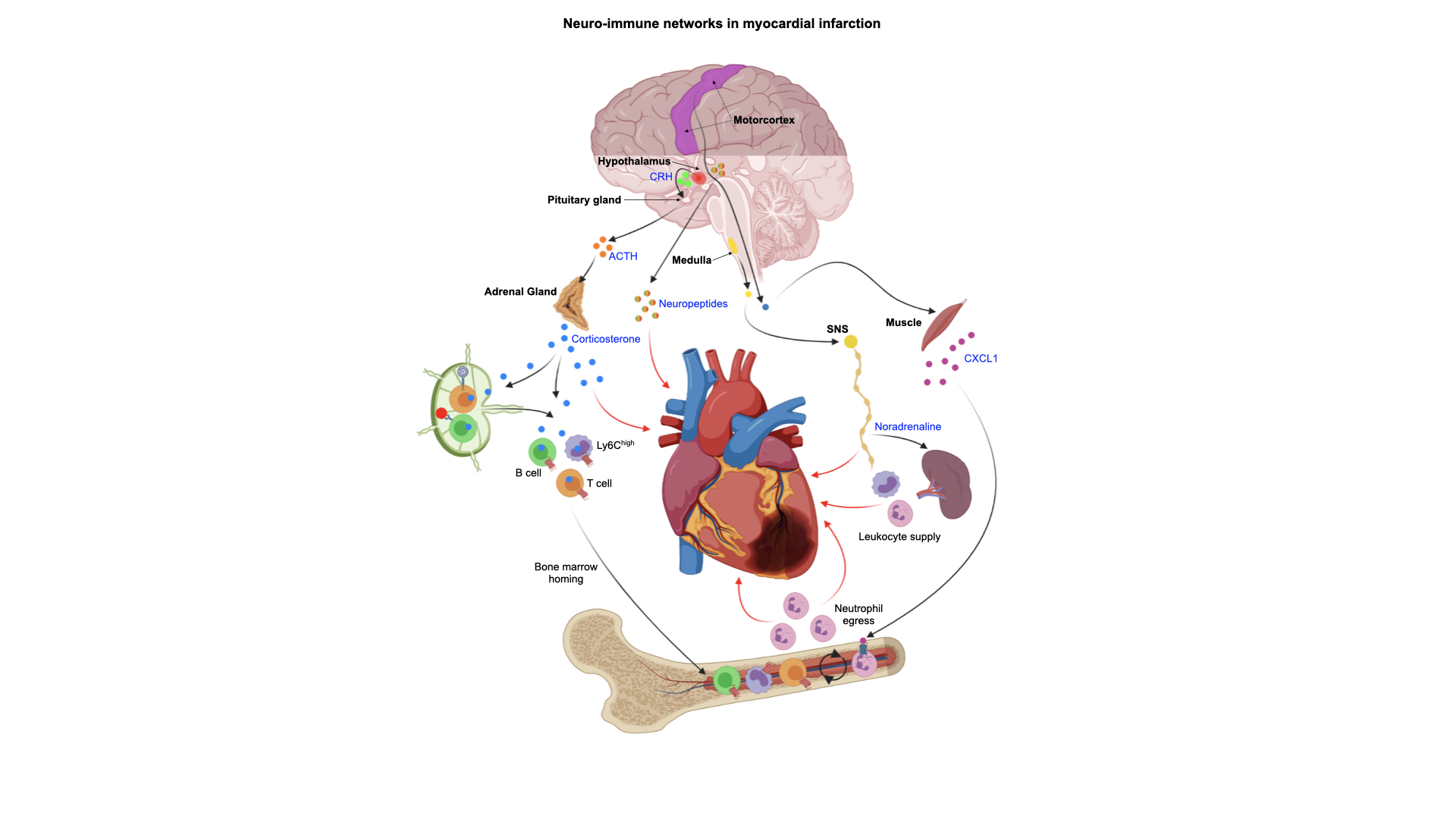
Myocardial infarction (MI) is a leading cause of mortality worldwide and is often followed by poor prognosis, with many patients progressing to ischemic heart failure. Beyond cardiovascular damage, acute MI is frequently accompanied by profound psychological disturbances, including elevated stress levels and increased susceptibility to depression. The immune system plays a central role in post-MI healing, and emerging evidence suggests that neuroimmune interactions may influence disease progression.
Research Question
Previous studies suggest that distinct brain circuits mediate large-scale leukocyte redistribution and functional changes during acute stress. However, it remains unclear whether and how these brain circuits regulate peripheral immune responses following MI.
Methods
We utilized the iDISCO-ClearMap pipeline to generate whole-brain activity maps in a murine model of MI at days 1, 3, 7, and 14 post-infarction, focusing on the spatial and temporal dynamics of cFOS–positive cells across brain regions. Neuronal activation will be validated using fiber photometry. To explore causal relationships, we conducted chemogenetic manipulations in CRH-Cre mice, including Cre-dependent expression of caspase-3 and hM3D(Gq) viruses to either ablate or activate CRH neurons, respectively. Cardiac function was assessed via echocardiography, and flow cytometry was used to evaluate immune cell populations in the infarcted heart.
Results
iDISCO-ClearMap analysis revealed robust activation of the central amygdala, locus coeruleus, rostral ventrolateral medulla, and CRH neurons within the paraventricular hypothalamus (PVH) following MI. Our preliminary data show that chemogenetic ablation of PVH CRH neurons prior to an MI improves post-MI cardiac function and increases leukocytes in bone marrow and heart. On the contrary, we found that chemogenetic stimulation of PVH CRH neuron in the post-MI period worsens cardiac function and reduces cardiac leukocyte infiltration.
Conclusion
CRH neurons in the PVH appear to play a critical role in coordinating neuroimmune responses following MI.
