Background: Cardiac amyloidosis (CA) is an important differential diagnosis in patients with symptomatic heart failure (HF) and increased left ventricular (LV) wall thickness. Cardiac magnetic resonance (CMR) imaging is useful to differentiate the underlying causes of left ventricular hypertrophy and to quantify myocardial remodeling and function. The prognostic utility of CMR parameters in this context has not been investigated.
Methods: 112 patients aged ≥60 years with symptomatic HF (NYHA class II to IV), LV ejection fraction (EF) ≥40%, LV wall thickness ≥12mm, NT-proBNP ≥1000pg/mL and hs-troponin T ≥14ng/L were prospectively included. CMR, bone scintigraphy, screening for monoclonal protein and, if necessary, myocardial biopsies were performed in all patients to diagnose cardiac amyloidosis. We evaluated the pre-specified secondary endpoints all-cause mortality and HF hospitalization after 1 year. Patients were stratified based on the presence or absence of cardiac amyloidosis. Univariable Cox regression analysis of CMR parameters and cardiac biomarkers with outcome was performed.
Results: All-cause mortality and rate of HF hospitalization were similar between patients with amyloidosis and those with other causes of heart failure (Figures). 20% of amyloidosis patients and 24% of non-amyloidosis patients had died (p=0.524). 16% of patients with amyloidosis and 26% of patients without amyloidosis were hospitalized for heart failure (p=0.230). Higher extracellular volume fraction (hazard ratio (HR) of 1% increase: 1.051, 95% confidence interval 1.001-1.103, p=0.047), higher levels of Troponin T (HR of 1 pg/ml increase: 2.218 (1.215-4.046), p=0.009), NT-proBNP (HR of 1 pg/ml increase: 2.463 (1.591-3.509), p<0.001) and lower LVEF (HR of 1% decrease: 0.959 (0.927-0.992), p=0.015) at baseline were associated with mortality. There was no association of native T1 with either all-cause mortality or HF hospitalization.
Conclusion: One year event rate was similar between patients with cardiac amyloidosis and those with other causes of heart failure. Parameters of myocardial remodelling, LV function and troponin level were associated with outcomes, the strongest association was observed with NT-proBNP. These data show that the severity of heart failure determines the mid-term prognosis. Whether amyloid cardiomyopathy with specific treatment options may impact long-term outcome has to be investigated by further follow-up.
The trial is registered at clinicaltrials.gov, study ID NCT04862273.
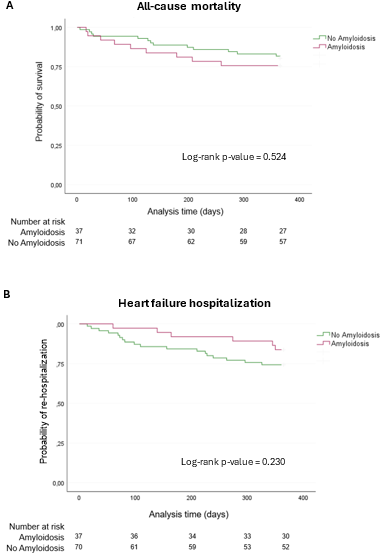
Figure: A, Kaplan–Meier survival curve showing the 1-year survival of patients with amyloidosis compared to those without amyloidosis. B, Kaplan–Meier curve illustrating the proportion of patients remaining free from heart failure hospitalization within one year in amyloidosis patients compared to non-amyloidosis patients.
