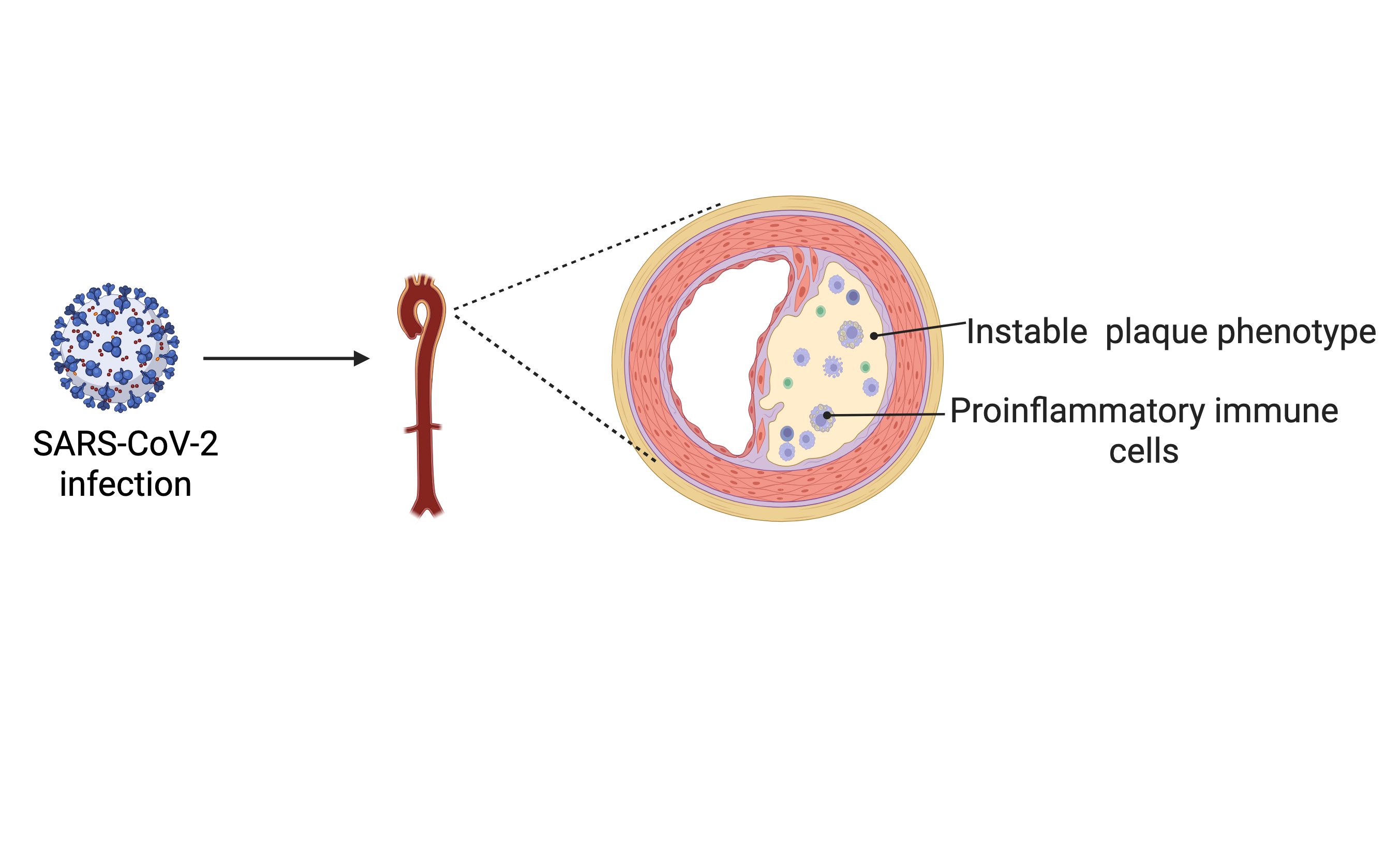
Background: Viral respiratory infections, including SARS-CoV-2, are associated with an elevated risk of major adverse cardiovascular events (MACE). Although effects of SARS-CoV-2 on the vasculature and atherosclerotic plaque development have been proposed, definitive in vivo insights on how SARS-CoV-2 affects atherosclerotic plaque development have been limited.
Methods: LDLR-/- mice were fed with a high-fat diet for 8 weeks to develop moderate atherosclerotic plaques in the aorta. Mice were then infected with 1000 plaque-forming units of a mouse-adapted SARS-CoV-2 strain compared to mock infection. Histology (H&E, Sirius Red) of aortic roots were performed 28 days after viral injury to analyse plaque size and stability (fibrous cap/necrotic core ratio). Single-cell RNA sequencing (scRNA-seq) of aortas was performed to characterise the composition and phenotypes of atherosclerotic plaques after viral injury. Differentially expressed gene-, and ligand-receptor analyses (CellChat) were used as readouts.
Results: In the aortic root, we observed increased features of plaque instability after SARS-CoV-2 infection (fibrous cap/necrotic core ratio, mock: 1.19±0,32 vs. SARS-CoV-2: 2,7±0,42, p=0.013). In contrast, overall plaque size remained unchanged, consistent to the development of an unstable plaque phenotype. To gain molecular insights into how SARS-CoV-2 affects the composition and phenotype of atherosclerotic plaques, we performed scRNA-seq of plaques 28 days after SARS-CoV-2 infection. Here, we found a proinflammatory plaque phenotype characterised by enrichment of immune cells. Numbers of recruited Mhc-II+/Ccr2int macrophages (p=0.02) and dendritic cell (DC) subtypes (cDC1 p=0.02, CCR9+ DCs p=0.043, mature DCs: p=0.03) were increased, while resident macrophage numbers were unchanged. In addition, we also found increased T-cell subset numbers (Naive Cd4+ p=0.004, T-regs p=0.01, Naive Cd8+ p=0.0005, Il17+Cxcr6+ p=0.008). Investigating the transcriptomic profile of plaque immune cells revealed that proinflammatory cytokines were significantly upregulated in macrophages (Ccl2, Cxcl10, IL-1α), dendritic cells (Ccl5, Cxcl9), and T cells (TNF and TNF superfamily members). In addition, recently described immunoregulatory endothelial cells were induced, which express major histocompatibility complex class II genes and can present antigens to T cells, thereby facilitating their recruitment to the vessel wall. Ligand-receptor analysis revealed a significant upregulation (all pathways: p < 0.05 after differential expression testing) of various proatherogenic pathways, indicating a complex SARS-CoV-2-induced communication network. Here, endothelial cells showed increased recruitment signalling to macrophages and T-cells via the CXCL12-CXCR4 axis. In addition, we found complex cellular communication between immune and stromal cells, where macrophages communicated with fibroblasts via PDGF and TNF-related pathways, as well as through paracrine mechanisms mediated by CCL pathways. T-cells communicated with fibroblasts and endothelial cells via TGF-beta-related pathways.
Conclusion: Our data, for the first time, dissects the in vivo effects of SARS-CoV-2 on the atherosclerotic vasculature, providing mechanistic insights into how SARS-CoV-2 infection affects the instability of atherosclerotic plaques.