Background:
Progressive loss of left atrial compliance, increased atrial pressures, and consequent pulmonary vascular remodelling are well-recognised features of disease progression in patients with heart failure and a preserved ejection fraction (HFpEF) and atrial fibrillation. However, there is a lock of comprehensive invasive hemodynamic data in this population. Here, we present a technique that integrates echocardiography and invasive pressure measurements in patients undergoing pulmonary vein (PVI) isolation to estimate atrial pressure-strain loops and atrial compliance.
Methods:
We retrospectively included patients with symptomatic, paroxysmal atrial fibrillation and preserved ejection fraction who underwent PVI at a single center in Berlin between March and July 2023. Inclusion criteria were sinus rhythm at admission, left atrial pressure measurements and echocardiographic left atrial strain measurements acquired within 24 hours of the procedure. From these data, we derived left atrial pressure-strain loops to estimate atrial compliance. Patients were categorized as non-failing and HFpEF, defined by the presence typical signs and symptoms and elevated NT-proBNP levels (>220 pg/mL).
Results:
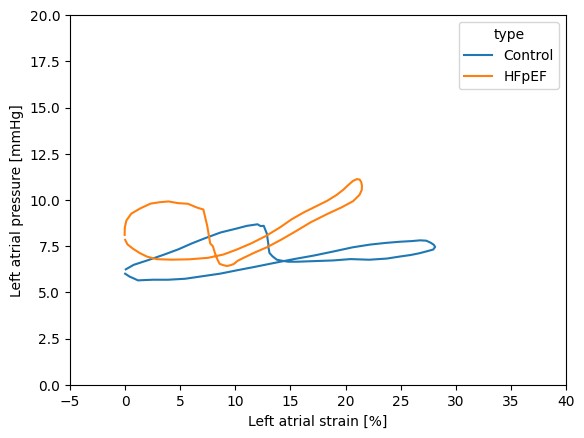
Among 29 patients, left atrial pressure-strain loops were successfully reconstructed in 21 (11 without and 10 with HFpEF). Patients with HFpEF were older (66.9 vs. 58.18 years) and had increased levels of NT-proBNP (637.6 vs. 80.0; pg/ml), while ejection fraction (61.7% vs. 62.7%), BMI (27.0 vs. 28.5 kg/m²) and left atrial volume index (41.63 vs. 37.62 ml/m²) were similar. Patients with HFpEF had numerically reduced left atrial reservoir strain (23.53 vs 28.01; %; p = 0.14) and increased maximum atrial filling pressures (maximum pressure of the atrial v-wave; 15.19 vs 9.18; mmHg; p < 0.01). Estimated atrial compliance during passive filling (reservoir strain / v-wave peak height) was decreased in HFpEF patients (3.27 vs 7.10; %/mmHg, p < 0.001)
Conclusion:
In patients undergoing PVI, the integration of echocardiography and invasive pressure data enables reconstruction of atrial pressure–strain loops and estimation of atrial compliance. In atrial fibrillation patients with HFpEF, atrial compliance was markedly reduced. Future studies are warranted to determine whether this parameter predicts clinical outcomes.
Funding: Funded by DFG projects 497794268, 437531118 and SFB 1470 subproject B01.