Background: Valvular integrity, rhythm stability, and hemodynamic performance are core determinants of cardiac status. Their timely appraisal is currently hindered by fragmented, expert‑dependent workflows: separate auscultation, ECG, and echocardiographic measurements. This fragmentation increases handoffs, prolongs bedside evaluation, and consumes limited clinical resources. A single, short, synchronized recording could streamline initial screening and reduce handoffs.
Objective: A novel, single wireless system capturing synchronous electrical, acoustic, mechanical, and impedance signals could enable integrated screening and quantitative assessment in one brief session. To evaluate the diagnostic performance of such a system, a multimodal patch device (ECG, impedance cardiography, phonocardiography, seismocardiography, bioimpedance spectroscopy) was compared to expert findings and echocardiography for (1) the classification of pathologic murmurs aortic associated with aortic valve stenosis (AS), (2) the detection of atrial fibrillation/flutter (AF/AFl), and (3) the estimation of stroke volume (SV).
Methods: Adults (n=1.095) were consecutively enrolled on cardiology wards (n=545, routine echocardiography) and surgery wards (n=550, pre-operative evaluation). The patch system recorded ~3-minute synchronous signals at the bedside. The patients underwent a cardiologic examination in parallel (including 12-lead ECG and echocardiography), unaware of the results of the patch system.
Supervised deep learning was applied to multi‑modal patch signals for valve, rhythm, and hemodynamic tasks. For the AS classification, a ResNet‑18 model, trained on a publicly available dataset (~5k recordings) was evaluated with patient‑level 4‑fold stratified cross‑validation scheme on the PCG signals of a representative subset (58 patients) of the study cohort. Arrhythmia detection employed a deep-learning model pre‑trained on public 12‑lead ECG datasets (~45k records), finetuned to the integrated system’s lead configuration and evaluated on a hold‑out test (211 patients) randomly sampled from the study cohort. SV estimation combined impedance and seismocardiographic features; performance was evaluated in a subset of 206 patients versus echocardiographic SV.
Results: AS classification achieved a sensitivity of 78.6%, and specificity of 85.6%. AF/AFl detection reached an sensitivity of 87.5%, specificity of 96.0%, and AUC of 0.988. SV estimation showed root mean squared error of 15.0 ml relative to echocardiography. These findings indicate that simultaneous valve, rhythm, and hemodynamic assessment is feasible from one short, synchronized recording.
Conclusion: A short, bedside recording with a wearable, wireless multimodal patch yielded preliminary performance across valvular heart disease classification, AF/AFl detection, and SV estimation when compared with expert and echocardiographic references. This integrated approach may streamline early cardiac screening and reallocate specialist time to higher-value decision making. However, larger, demographically characterized, externally validated cohorts and comprehensive safety monitoring are required before routine clinical adoption.
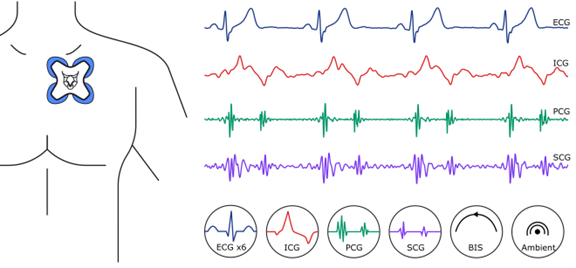
Fig 1: (Left) Wireless sensor patch placement. (Right) Signals collected synchronously by the system.
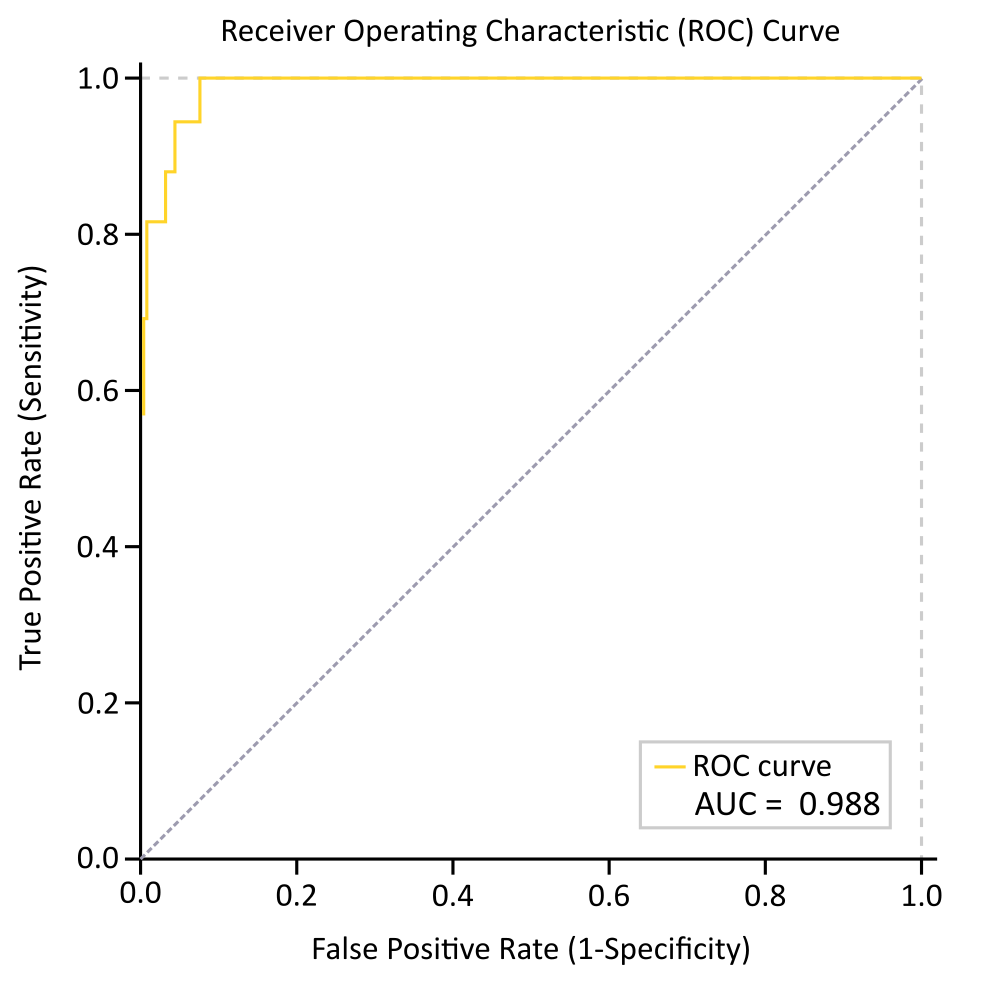
Fig 2: Receiver operating characteristic for the AF/AFl detection task of the ResNet-18 model on the clinical investigation data subset.
