Background: Cardiac troponin is a biomarker for myocardial injury and is integral to risk prediction for incident heart failure and to prognosis after mycardical infarction. In acute heart failure (AHF), however, extensive myocardial necrosis is not invariably present. In some presentations, volume overload with consecutive myocardial wall stress is the dominant driver of decompensation. Whereas troponin T predominantly reflects overt myocardial damage, high-sensitivity troponin I (hsTnI) can detect subtle injury associated with myocardial strain.
Aims: To evaluate the distribution of hsTnI and to investigate whether hsTnI levels can predict outcomes in heart failure-related AHF.
Methods: Adults hospitalised with AHF were prospectively enrolled from 2019 to 2024 in an oberservational, single-center cohort study. Patients undergoing percutaneous coronary intervention at the index hospitalization were excluded. HsTnI levels were categorised as high (> 99th percentile) versus low (≤ 99th percentile). Associations between hsTnI category and the composite endpoint of rehospitalisation for heart failure or death during the follow-up period were evaluated using Cox proportional-hazard models adjusted for age, sex and creatinine at baseline. Two-sided p<0.05 was considered statistically significant.
Results: 135 AHF patients were included. Median age was 70 years (IQR 58, 78), 52 patients (38.5%) were female. Common triggers of AHF were tachycardia (n=41, 32%), infection / sepsis (n=21, 16.4%) and hypertensive crisis (n=14, 10.9%). Median baseline hsTnI in the overall cohort was 41 (IQR 20, 144) pg/ml. At baseline, high hsTnI was observed in 61 patients (45%) with a median hsTnI of 157.0 (IQR 73.3, 514.7) pg/ml. Low hsTnI at baseline was observed in 74 patients (55%) with a median hsTnI of 21.5 (IQR 11.9, 33.1) pg/ml. High baseline hsTnI was associated with higher baseline NT-proBNP [11708 (IQR 6544, 20104) ng/l versus 4680 (IQR 2446, 9857) ng/l]. More patients with high basline hsTnI required treatment on an intensive care unit [n=30 (49.2%) versus n=14 (19.2%), p=0.00039] (Figure 1). Overall, 59 patients experienced rehospitalisation for heart failure or death within a median follow-up time of 2.13 years (95% CI 1.73, 3.28). 3-year mortality rate was 54.85%. High baseline hsTnI was associated with an increased risk of the composite endpoint [adjusted HR 1.85 (95% CI 1.0, 3.4), p=0.048], while high discharge hsTnI was not associated with the endpoint (Figure 1).
Conclusion: High hsTnI at presentation in heart failure-related AHF indicated higher volume load. HsTnI can help to predict in-hospital trajectories and long-term outcomes in AHF. Further analyses across heart failure phenotypes are warranted to refine thresholds and optimise clinical management.
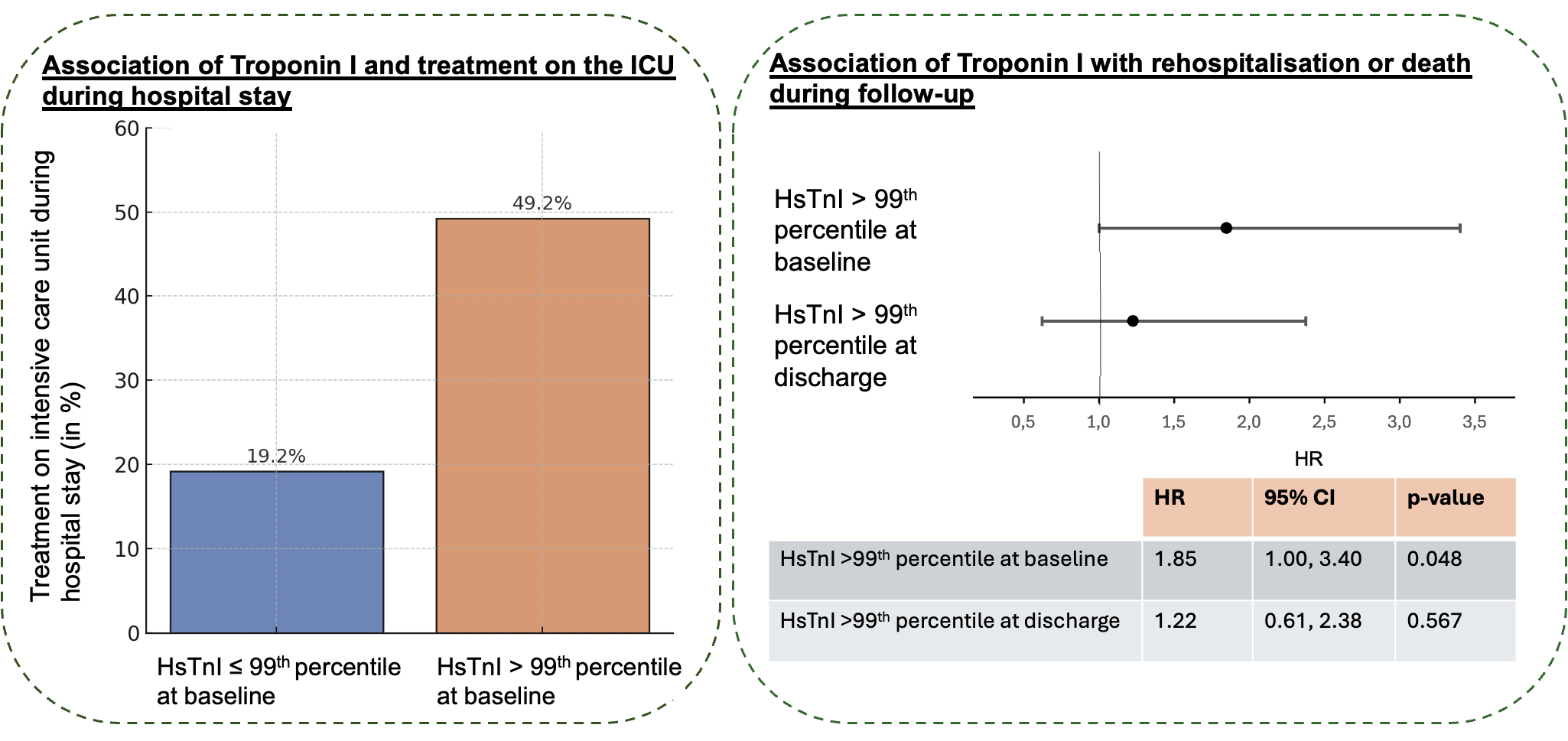
HsTnI, high-sensitivity Troponin I; ICU, intensive care unit; HR, hazard ratio; CI, confidence interval
