Background: Inflammation plays a key role in the development and progression of heart failure by promoting maladaptive cardiac remodeling, impairing myocardial contractility, and driving fibrosis. T2 mapping by cardiovascular magnetic resonance (CMR) enables non-invasive quantification of myocardial inflammation and has shown prognostic value in selected disease entities such as acute myocarditis, aortic stenosis, and cardiomyopathies. However, large-scale data from unselected patient cohorts are lacking.
Purpose: To assess the prognostic value of T2 mapping in a large, real-world cohort undergoing clinical CMR.
Methods: Patients were enrolled from the prospective, observational BioCVI registry. Septal T2 values were measured on basal or mid-ventricular short-axis slices with optimal image quality. The primary endpoint was a composite of all-cause mortality, aborted sudden cardiac death, and hospitalization for heart failure at 1-year follow-up.
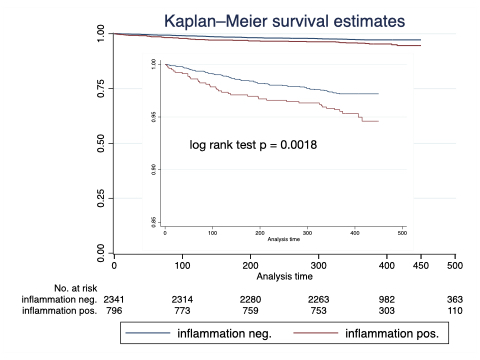
Results: A total of 3,852 patients were recruited between April 2017 and October 2025; complete follow-up was available in 3,137 patients. The primary endpoint occurred in 112 individuals (3.6%). Patients reaching the endpoint had significantly higher T2 values, lower left ventricular ejection fraction (LVEF), and higher NT-proBNP levels. Each 1-ms increase in T2 was associated with a hazard ratio (HR) of 1.10 (95% CI 1.04–1.16; p=0.0008). In multivariable Cox regression including LVEF, end-systolic volume index, NT-proBNP, estimated glomerular filtration rate, age, NYHA class, and other clinical variables, T2 remained independently associated with the primary endpoint. Adding T2 to the clinical model improved prognostic performance (likelihood ratio test χ²=4.8; p=0.041). Using a cutoff of 40 ms, patients with elevated T2 had significantly worse outcomes.
Conclusion: In a large, unselected clinical CMR cohort, myocardial T2 mapping independently predicted the combined endpoint of all-cause mortality, aborted sudden cardiac death, and heart failure hospitalization. These findings underline the central role of myocardial inflammation in heart failure development and highlight the prognostic value of incorporating T2 mapping into routine clinical assessment.