Introduction
Atrial fibrillation (AF) has markedly increased in prevalence, underscoring the need to elucidate its underlying mechanisms. Impaired calcium handling contributes to AF-associated electrical instability. We identified reduced miR-144 levels in AF patient samples, and NOS1AP—a regulator of SERCA activity—was predicted as a miR-144 target, suggesting a mechanistic link between miR-144 and AF.
Objective
This study aimed to elucidate the molecular mechanisms by which miR-144 contributes to the development of AF.
Methods
Mice were treated with a miR-144 inhibitor, and a miR-144 knockout model was generated. Echocardiography and in vivo electrophysiological were performed to study cardiac dimensions and function as well as susceptibility for atrial arrhythmias, respectively. Luciferase reporter assays and gene/protein expression analyses were performed to confirm NOS1AP as target. Atrial cardiomyocytes were isolated from miR-144 knockout mice for patch-clamp and calcium transient measurements.
Results
MiR-144 inhibition and knockout increased atrial tachycardia inducibility and caused left atrial dilatation without affecting global cardiac function. NOS1AP was validated as a direct miR-144 target, showing increased atrial expression in knockout mice. Patch-clamp analyses revealed reduced calcium currents and intracellular calcium transients in miR-144-deficient cardiomyocytes.
Conclusion
These findings suggest that loss of miR-144 causes NOS1AP upregulation which results in impaired SERCA-mediated calcium reuptake leading to atrial arrhythmogenesis. Additional studies are warranted to further investigate this novel mechanism and the therapeutic potential of targeting the miR-144/NOS1AP axis in AF.
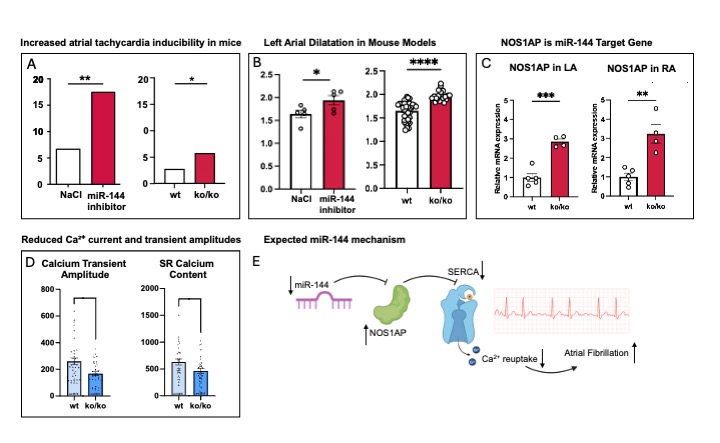
Figure 1 MiR-144 inhibitor and knockout mice show an increased inducibility of atrial tachycardia (episodes >1s with a cycle length <100ms; Chi-square test; inhibitor group p < 0.01(**), knockout group p < 0.05(*); n = 10 wt, 4 ko mice)(A) and a left atrial dilatation on echocardiographic assessment (n = 5 each; one-way ANOVA with Tukey’s multiple comparisons test; inhibitor group p < 0.05(*), knockout group p < 0.0001(****)).(B) Gene expression analyses and luciferase reporter assays identified NOS1AP as a target of miR-144 in knockout mice (mean ± SEM, p<0.001(***), p<0.01(**) unpaired t-test).(C) In atrial cardiomyocytes from knockout mice, calcium transient amplitudes and SR calcium content were significantly reduced, indicating impaired SERCA-mediated calcium reuptake. (mean ± SEM; p < 0.05, Mann–Whitney test; SR calcium load quantified by Ca²⁺ transient amplitude, mean ± SEM, p < 0.05, Mann–Whitney test)(D) The schematic model illustrates the proposed mechanism by which loss of miR-144 leads to NOS1AP upregulation, reduced SERCA activity, disturbed calcium homeostasis, and subsequently increased atrial fibrillation.(E)
